Bromination of Safrole
Reagents
100 mmol (16.21g) safrole
210 mmol (21.58g) NaBr
190 mmol (19.00g/10.33mL 98% conc.) H2SO4
40mL (38.43g) GAA (solvent)
Procedure
The reaction flask was charged with H2SO4 (190mmol) and chilled to 0C. The GAA was added as such as rate that the temperature was kept below 5C. The NaBr was then added portion-wise over five minutes. The flask took on a tangerine orange appearance. Once the addition was complete (exothermic), the safrole was added at a rate which maintained the previous temperature. After the addition a brown/green color was noted, along with a thin deposit of what was assumed to be NaBr on the sides of the flask. Noxious fumes were evolved, the author would suggest sniffing them to get high. The flask was left to stir on an ice bath for 24 hours.
Note: the safrole was not purifed or distilled from the sassafras oil. According to the original author the phenolic compounds in the oil help to quench free radical side reactions. The oil known to be 89% safrole.
3 hrs - the flask has progressed to a black/purple color
Reagents
100 mmol (16.21g) safrole
210 mmol (21.58g) NaBr
190 mmol (19.00g/10.33mL 98% conc.) H2SO4
40mL (38.43g) GAA (solvent)
Procedure
The reaction flask was charged with H2SO4 (190mmol) and chilled to 0C. The GAA was added as such as rate that the temperature was kept below 5C. The NaBr was then added portion-wise over five minutes. The flask took on a tangerine orange appearance. Once the addition was complete (exothermic), the safrole was added at a rate which maintained the previous temperature. After the addition a brown/green color was noted, along with a thin deposit of what was assumed to be NaBr on the sides of the flask. Noxious fumes were evolved, the author would suggest sniffing them to get high. The flask was left to stir on an ice bath for 24 hours.
Note: the safrole was not purifed or distilled from the sassafras oil. According to the original author the phenolic compounds in the oil help to quench free radical side reactions. The oil known to be 89% safrole.
3 hrs - the flask has progressed to a black/purple color




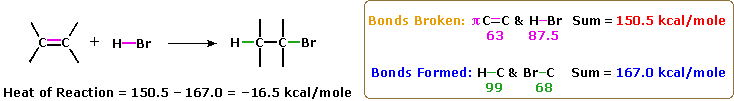





 Some more examples of substitution-elimination reactions in the chemical literature originally provided by another staff member of the Hive:
Some more examples of substitution-elimination reactions in the chemical literature originally provided by another staff member of the Hive:



