
Drying tubes are small tubes with a connection at one end. To be used, they are filled with a drying agent, such as Drierite (calcium sulfate, anhydrous). To prevent the particles of drying agent from falling out of the drying tube, they are lightly plugged at both ends with glass wool. The drying tubes used in the organic chemistry teaching labs can be connected to the 14/20 standard taper glassware by connecting them first to a thermometer adaptor.
Drying tubes are used in organic reactions to prevent moisture from the air from entering a reaction flask. They must always be packed loosely, both the glass wool and the drying agent, because they provide a path for the release of pressure which can build up during a reaction. In other words, if they are so tightly packed that air cannot flow through them, when placed in an apparatus they will seal the system. You should never have a closed system during an organic reaction, since if enough pressure builds up, it will explode.
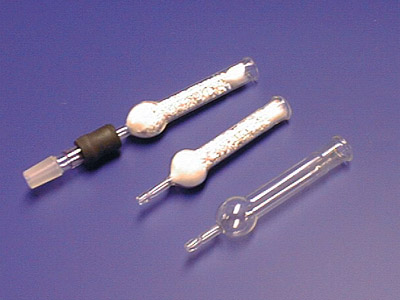
Shown below are three drying tubes. The lower one is a clean, empty drying tube; the middle one has been plugged with glass wool and filled with drying agent. The top drying tube is connected to a thermometer adaptor, ready to be inserted in a 14/20 joint in a reaction set-up.
Location: Community glassware. When called for in a reaction, they will be placed in a bin on the TA's desk.