I believe that this is a new twist on an old discussion, and hope it's not entirely incorrect.
Maybe those of you who were tired of this particular type of thread five years ago will find something fresh to fume about here.
Just kidding. Or am I?
There's an interesting discussion on WD started by iknowit on a faster way to reduce PSE with phosphorous acid. It interested me because I started looking at the reaction in a different way than I had before, and several points have come up that lead me to wonder if any of the old explanations for this reaction are correct.
Every reference I've seen says that iodoephedrine is formed by the action of HI on ephedrine. However, it was noted long ago that an exothermic reaction between PSE and I2 takes place with no more provokation than stirring the mixture, which quickly melts. This mixture reduces in a reaction with RP just as expected. I can't imagine that anything other than the formation of iodoephedrine is taking place in what was observed, but if that's true then HI isn't needed for this part of it.
In researching the matter, one author that seemed to be talking about the same thing mentioned that an SN2 was ordinarily the way this kind of thing takes place, as did one of the posters to the WD threads. The carbon attached to the OH is highly acidic, the article said, and the OH, being highly basic, doesn't come off easily. However, a characteristic of SN2 is molecular inversion, since in this the halogen acid attacks from the back side. I'm no chemist but I don't see that a reversal takes place. But even if it does, what reaction takes place between PSE and I2 alone?
Another tidbit from the thread was the suggestion that using the HCl salt of PSE wastes HI, because it must be expended in freeing HCl from the molecule prior to the reduction. It was difficult to find a molecular model that showed PSE HCl, but I finally did find one, and it showed HCl bonded ambiguously to the whole molecule. I don't know if that's correct, but see no reason why HCl would bond with the carbon we want to manipulate, and as long as that's not the case, see no reason why the HCl needs to bother about it at all. Wouldn't it be fine, wherever it is, right up until the end of the reaction, at which point the mixture is based? It does make sense that at that point the base would be both removing the HCl as well as oxidizing the iodoamphetamine.
Then again, that last makes no sense because it would imply the entire reduction occurs when the mixture is based, and that sounds very unlikely. Could it be that this is one of the functions of the phosphorus, in whatever form it may be used, to oxidize the HI from the iodoephedrine,leaving a proton and completing the reduction?
My thinking is probably wrapped around the axle somewhere in this. Any insights are much appreciated!
Thanks,
PP
Maybe those of you who were tired of this particular type of thread five years ago will find something fresh to fume about here.

Just kidding. Or am I?

There's an interesting discussion on WD started by iknowit on a faster way to reduce PSE with phosphorous acid. It interested me because I started looking at the reaction in a different way than I had before, and several points have come up that lead me to wonder if any of the old explanations for this reaction are correct.
Every reference I've seen says that iodoephedrine is formed by the action of HI on ephedrine. However, it was noted long ago that an exothermic reaction between PSE and I2 takes place with no more provokation than stirring the mixture, which quickly melts. This mixture reduces in a reaction with RP just as expected. I can't imagine that anything other than the formation of iodoephedrine is taking place in what was observed, but if that's true then HI isn't needed for this part of it.
In researching the matter, one author that seemed to be talking about the same thing mentioned that an SN2 was ordinarily the way this kind of thing takes place, as did one of the posters to the WD threads. The carbon attached to the OH is highly acidic, the article said, and the OH, being highly basic, doesn't come off easily. However, a characteristic of SN2 is molecular inversion, since in this the halogen acid attacks from the back side. I'm no chemist but I don't see that a reversal takes place. But even if it does, what reaction takes place between PSE and I2 alone?
Another tidbit from the thread was the suggestion that using the HCl salt of PSE wastes HI, because it must be expended in freeing HCl from the molecule prior to the reduction. It was difficult to find a molecular model that showed PSE HCl, but I finally did find one, and it showed HCl bonded ambiguously to the whole molecule. I don't know if that's correct, but see no reason why HCl would bond with the carbon we want to manipulate, and as long as that's not the case, see no reason why the HCl needs to bother about it at all. Wouldn't it be fine, wherever it is, right up until the end of the reaction, at which point the mixture is based? It does make sense that at that point the base would be both removing the HCl as well as oxidizing the iodoamphetamine.
Then again, that last makes no sense because it would imply the entire reduction occurs when the mixture is based, and that sounds very unlikely. Could it be that this is one of the functions of the phosphorus, in whatever form it may be used, to oxidize the HI from the iodoephedrine,leaving a proton and completing the reduction?
My thinking is probably wrapped around the axle somewhere in this. Any insights are much appreciated!
Thanks,
PP






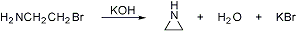
 More to come!
More to come!