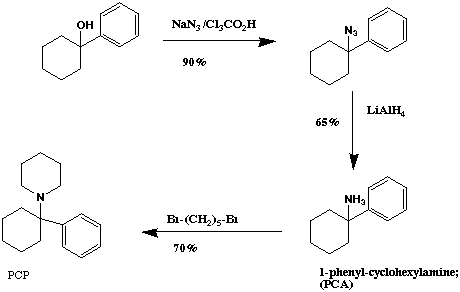
The next three routes use PCA (1-phenyl cyclohexylamine) as a precursor for either PCP or other analogs.
PCA itself is an active compound, and has been under clinical study as a potential anesthetic agent. It is approximately one half as potent than PCP and appears to have similar actions. PCA can be transformed into other PCP analogs by alkylation of the amino group by a number of methods. It is possible to react 1,5-dibromopentane with PCA in order to create PCP (yield 65-75% after 1-6 days; ref. 10, 11, 50-56). Reaction of PCA with formic acid and formaldehyde gives the N,N-dimethyl analog of PCP (ref. 10). An attractive procedure from the standpoint of clandestine synthesis would be the reductive alkylation of PCA with acetone and aluminum amalgam to give the potent analog N-isopropyl phenylcyclohexylamine.
The first method using PCA as an intermediate is Scheme IV. This route has received little attention among clandestine chemists, but has potential for synthesis of PCP derivatives without use of watched chemicals. This route gives an overall yield of ~40% with a difficulty rating of 7 out of 10, and a hazard rating of 8 out of 10 (ref. 64).

The PCOH starting material can be purchased or prepared by reaction of phenyllithium or phenyl grignard with cyclohexanone. This alcohol is then be transformed into the amine by reaction with sodium azide (~90% yield) followed by reduction. The azide is easily reduced to the final amine by a number of reagents, such as Raney Nickel, Pd/C, magnesium metal, lithium aluminum hydride, or sodium borohydride (ref. 50-54).
One of the better procedures for formation of the azide involves adding the alcohol to a suspension of sodium azide in trichloroacetic or trifluoroacetic acid (ref. 39). Another reagent that has been used to form the azide is diisopropylazodicarboxylate (ref. 55). It should be noted that sodim azide is highly toxic, and contact with acid will release the even more deadly hydrazoic acid.
This method is generally easy, and starts with inexpensive and commercially available 1-phenylcyclohexene, or alternately from1-phenylcyclohexanol (PCOH) (ref. 10, 56, 58, 65). PCOH or phenylcyclohexene is reacted with sodium cyanide and H2SO4 to give N-formyl PCA in about 50-60% yield. N-formyl PCA is readily hydrolyzed in either acid or base conditions to give PCA, which can then be alkylated to give PCP and other analogs, as illustrated in Scheme IV. This route has an overall yield of ~30% with a difficulty rating of 1-2 out of 10, and a hazard rating of 3 out of 10 (ref. 64).
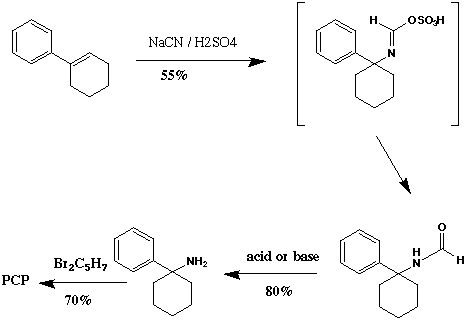
Procedure (ref. 11):
Preparation of PCA: To a mixture of 15.8 gm of 1-phenylcyclohexene (0.1 mole) and 12.2 gm of NaCN (0.25 mole) in 50 mL of dibutyl ether, was added 30 mL of H2SO4 over 1 hr. After stirring for an additional 1 hr, the reaction mixture was poured into water and extracted with ether. The solvents were removed in vacuo, 30 mL of HCl was added to the residue, and the mixture refluxed for 3 hrs. After cooling, the aqueous layer was seperated, basified with NaOH, and extracted with ether. The HCl salt was prepared by adding dry HCl in isopropanol, followed by evaporation. 20 mL of acetone was then added to the residue, followed by recrystallization twice from methanol/ether to give needles (mp 247-248 C).
Preparation of PCP from PCA: A mixture of 8.69 gm of PCA, 11.5 gm of 1,5-dibromopentane, and 8.0 gm of anhydrous K2CO3 in 50 mL of dry DMF was stirred and heated. At 50-55 C an exothermic reaction took place and the temperature rose to 95-100 C. The flask was heated for 1 hr on a steam bath, poured into ice cold water and extracted with ether, followed by distillation and recrystallization to give the final compound.
This reaction starts with low cost materials, but the procedure is more lengthy, difficult, and dangerous than previous methods. Overall yield is around 30%, with a difficulty rating of 9 out of 10, and a hazard rating of 8 out of 10 (ref. 64, 10).
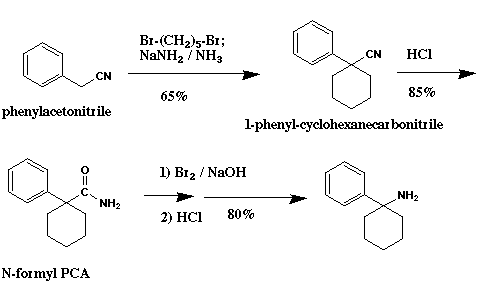
The starting material is phenylacetonitrile (benzyl cyanide), which is a watched precursor for methamphetamine synthesis. It can, however be easily synthesized from benzyl chloride and NaCN. Benzyl chloride can also be prepared without much difficulty, by the chlorination of toluene. Also certain (unwatched) substituted phenylacetonitriles could be used to yield potent PCP analogs, e.g. 3-methoxyphenylacetonitrile.
Chemistry: In the first step, phenylacetonitrile is reacted with 1,5-dibromopentane and strong base to yield 1-phenyl-1-cyclohexanecarbonitrile. The base can be either NaNH2 (sodamide; 65% yield) or sodium hydride in anhydrous DMSO (better). If NaNH2 is used it may be purchased commercially or prepared from sodium metal and liquid ammonia (-30 C).
In the next step, the carbonitrile is hydrolyzed with either HCl or H2SO4 in trifluoroacetic acid to give 1-phenyl-1-cyclohexanecarboxamide (yield 85%; ref. 10). Some substituted phenylacetonitriles are extremely resistant to acid hydrolysis, in which case basic hydrolysis may be more efficient (ref. 62).
Finally, the amide is subjected to a Beckmann rearrangement, giving N-formyl PCA, and the formyl group is hydrolyzed, to yield PCA in about 80% yield (ref. 63, 56). The PCA can be transformed into PCP or other analog as illustrated in Scheme V.
Procedure.
Step 1: 1-phenylcyclohexanecarbonitrile: A mixture of 222 gm (1.9 moles) of phenylacetonitrile and 400 gm (1.71 moles) of 1,5-dibromopentane was added over 6 hrs. to a refluxing suspension of 593 gm (7.54 moles) of sodamide in 4.5 L of ether. Reflux was continued overnight, followed by cooling in an ice bath and addition of 1.6 L of water over a period of 2.5 hrs, keeping the temperature at 21-24 C. The mixture was then refluxed for 1 hr. and cooled. Following filtration and seperation of the aqueous layer, the ether layer was washed with 1 L of water and 1 L of 3 N HCl. The solvent was then distilled to give 234 (72% yield) of crude product. Purification by fractional distillation (102 C / 0.1 mm Hg) gave 62% of pure nitrile.
Step 2: 1-phenylcyclohexanecarboxamide: A mixture of 130 g (0.7 mole) of 1-phenylcyclohexanecarbonitrile, 415 g of trifluoroacetic acid, and 59.5 g of H2SO4 was heated at reflux for 16 hrs., and then poured onto 500 gm of ice. A tan precipitate will form, which is washed with water, saturated aqueous Na2CO3, and then water again until neutral. The crude product weighed 121 gm (85.5% yield), and recrystallization from isooctane or hexane gives 72.3 gm of amide (50.8% yield), mp 85-88 C.
Step 3: 1-phenylcyclohexylamine (PCA): To a cooled solution of 198 gm of KOH in 1 L of water was added 18 mL of bromine, and the solution further cooled to 7 C. 67.3 gm (0.331 mole) of 1-phenylcyclohexanecarboxamide was added at once, and the mixture held at 3-9 C for 90 min. with stirring. It was then extracted 3 times with 200 mL of ether, 300 mL of concentrated HCl was heated to 50 C, and the extracts added. The mixture was heated to 110 C for 90 min., cooled to 85 C, and 210 gm of NaOH in 900 mL of water was added over 15 min. After cooling and extracting with 600 mL of ether, the solution was dried over NaOH pellets and the ether distilled to give a residue of crude amine (46.4 gm, 80.3% yield).
Next Section: PCP via N-benzoyl piperidine Back to Contents