Mining the web and following up leads in related articles , I came upon a couple of studies dealing with the reduction of arene aliphatic alcohols, checked the archives and found no listings on the HIve and so I present them here now for discussion.......java
Chlorotrimethylsilane/SodiumIodide/Zinc as a Simple and Convenient Reducing System:One pot Deoxygenation of Alcohols and ethersTsuoshi Morita, Yoshiki Okamoto, Hiroshi sakuraiSynthesis 32, 1981
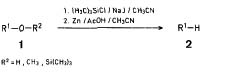 Note
Note: with this method both primary and secondary alcohols are reduced to hydrocarbons
Synthetic Method and Reactions;47. AlCl/Pd-Catalyzed Reduction of Aryl Substituded Alcohols and Olefins by Hydrogen Transfer from Cyclohexene
George A. Olah ; G.K. Surya Prakash
Synthesis 397, 1978

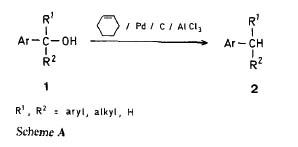
Note: with this method it seems only benzylic alcohols are reduced, might be a good way to reduce Pseudo-ephedrine......
Edited.....by java