When thinking about the auto-oxidation of olefins as isosafrole, anethole and/or isoeugenol, you have to imagine some sort of intermediary complex between a molecule of O
2 and the olefin double bond. There will be an
oxidative cleavage of your olefin, which in case of isosafrole will yield piperonal (anethole -> anisaldehyde, isoeugenol -> vanillin) and acetaldehyde.
In the past, I have tried some Na
2Cr
2O
7 and KMnO
4 mediated oxidations of isosafrole, isoeugenole, anethole and asaron. In case of isoeugenol and anethole, I have had performed a GC/MS analysis as well, and I found that in both cases, their corresponding benzaldehydes were present as the major substances. However, other concomitant substances were the original starting product and also their corresponding P2Ps! At the time, I was rather surprised as I didn't expect the latter to show up. However, I have no idea for how much their P2P analogues contributed to the composition of the final product: 5%? 10%? Anyway, not sufficient to isolate them for further synthetical needs. So, to summarize, auto-oxidation of anethole and isoeugenol does result in 4-methoxy-P2P and 4-hydroxy-3-methoxy-P2P, albeit in small amounts.
And all the sudden, there is a group of crazy Japanese writing a possibly interesting article in a language the majority of the Bees unfortunately doesn't understand. It seems that bubbling oxygen in a solution of olefin in DMSO results in the P2P analogue as the major product and the benzaldehyde as the minor one. This, however, does not fully agree with the theoretical principle of the olefin auto-oxidation. So, might there be something else going on?
No... and yes. At least, that is
my theory. My theory goes as follows:
You dissolve for instance a certain amount of isosafrole in DMSO and introduce a stream of oxygen into the mixture. The oxygen will form an intermediary complex with isosafrole, which will decompose into piperonal and acetaldehyde. These two aldehydes, however, are subject to auto-oxidation as well! In
Post 415088
(GC_MS: "The synthesis of peracids", Chemistry Discourse), I describe how perbenzoic acid can be formed by introducing oxygen in a solution of benzaldehyde in acetone. Acetaldehyde will result in peracetic acid while piperonal will yield 3,4-methylenedioxy perbenzoic acid. The actions of peracids on alkenes as isosafrole is (or should be) well known. No? Well, you might want to read
some pages on Rh's site...
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/peracid.html)
And
this is where the fun starts

. The epoxide (formed by the reaction between isosafrole and peracid) can rearrange to a zwitterion (positive charge on C1 and negative charge on O of the side chain), which on itself is a substrate in a Pfitzner-Moffatt oxidation reaction.
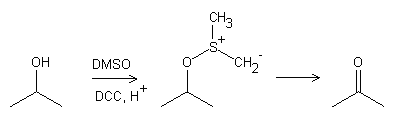
And voila, there you have your MD-P2P. I must add that the zwitterion I just talked about can also rearrange to MD-P2P spontaneously.
So, how did I pull this theory out of my ass all the sudden? The abstract mentions the presence of epoxidated alkanes, benzaldehydes etc if they used DMI, DMAc or DMF as solvent. Only when they used DMSO, there was a major presence of the P2P analogue. To me, it seems that DMI, DMAc and DMF allow the auto-oxidation to happen "the normal way". DMSO "rearranges" the equilibria of the side-reactions and oxidizes an intermediary zwitterion, yielding the beloved MD-P2P.