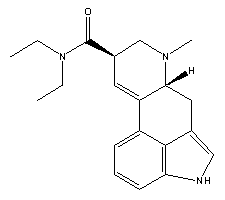
 Preparation of Lysergic Acid Diethylamide with Thionyl Chlorideby Protium and Vibrating_Lights
Preparation of Lysergic Acid Diethylamide with Thionyl Chlorideby Protium and Vibrating_Lights
ABSTRACT:Many methods have been explored in the preparation of lsysergic acid diethylamide from lysergic acid. The amide to be prepared is a disubstitued amide. That is, two of the hydrogens on the amide (-NH2 group) have been replaced with ethyl groups. Amides cannot be prepared directly by mixing a carboxylic acid with an amine. If the acid were to be simply mixed with the amine, the conjugate base of the acid would be produced, which would not react further while in solution. If the amine salt is isolated as a crystalline solid and strongly heated, the amide can then be prepared, however, this method would not be convienient due to the high temperatures required for the reaction. Thus, the most common route in amide preparation is through the intermediate acid chloride. Many different reagents have been proposed for the conversion to the acid chloride, but we believe that a detailed procedure using thionyl chloride has been previously unreported to the clandestine community. Herein we report a detailed procedure for the small scale preparation of lysergic acid diethylamide from lysergic acid and diethylamine, using thionyl chloride.MATERIALS:Hotplate/Stirrer combination
Erlenmeyer flask
Separatory funnel
Beaker
100 mL RB flask
Claisen head with fitting rubber septum
Graduated pipet
Syringe
Reflux Condenser
Thermometer adapter
Glass tubing
Rubber tubing
Test tube
Moist cotton
Clamps & Stands
3.57 g Lysergic acid
2.0 mL Thionyl chloride
4.5 mL Diethylamine
Ether (anhydrous)
10% aq. NaOH
10% aq. HCl
Na
2SO
4ASSEMBLY: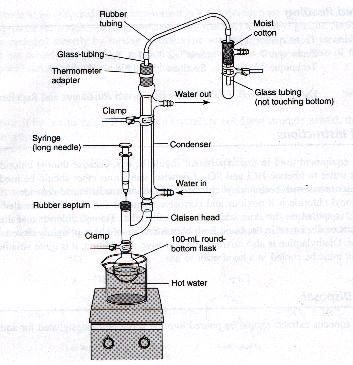
Assemble the apparatus as shown above in a darkroom.
In the presence of light, water can add to the double bond between the position of carboxide attachment and the aromatic ring.TIHKAL#26 You can omit the syringe for now. The gas trap shown in the figure will trap the evolution of hydrogen chloride and sulfur dioxide gases. Warm up the water in the beaker on the hotplate to about 90*C. Be careful not to boil the water and let the vapor come into contact with the reagents.
MECHANICS: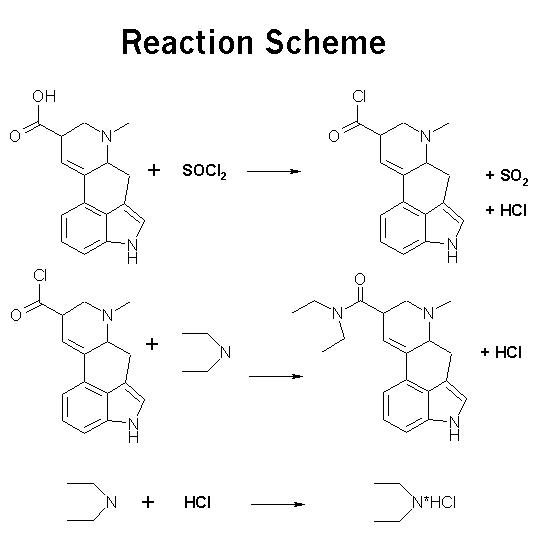 PROCEDURE :Preparation of the Acid Chloride:
PROCEDURE :Preparation of the Acid Chloride:Place 3.57 g of lysergic acid into a dry 100 mL roundbottom flask. Measure 2.0 mL of thionyl chloride into a graduated pipet, and removing the rubber septum, transfer the thionyl chloride into the flask. Replace the septum. Make sure that the thionyl chloride does not come into contact with either your skin, or the water in the bath. Make sure the equipment used is dry.
Begin to stir the mixture lightly, begin circulation of water into the reflux condenser, and gradually heat the mixture to reflux for 30 minutes.
Preparation of the Amide:Raise the apparatus out of the water bath and allow it to cool down to room temperature. After the mixture has cooled, remove the rubber septum and pour 25 mL of anhydrous ether into the reaction flask. Reattach the septum. Swirl this mixture until a homogenous solution is obtained.
Remove 4.5 mL of ice cold diethylamine and place it into a small erelynmeyer for storage. Add 8 mL of anhydrous ether to the amine in the flask. Keep the flask cool while you obtain a syringe.
Draw up the diethylamine/ether solution into a dry syringe and insert the needle through the rubber septum of the apparatus. Add the solution dropwise over a 10-15 minute period into the roundbottom flask. At this point you should notice a cloud of diethylamine hydrochloride forming in the flask. Set the mixture to stir for about 90 minutes.
Remove the rubber septum and pour 14 mL of a 10% aqueous sodium hydroxide solution into the flask in small portions.
Be certain that the concentration is not any higher than this, as the position of the carboxide attachment is very sensitive to acidic and basic pH.TIHKAL#26 Swirl occasionally or set to stir for 15 minutes. The sodium hydroxide should convert most of the remaining acid chloride to the sodium salt of the acid. This salt will be soluble in the aqueous layer. The diethylamine hydrochloride is also water soluble. Any remaining thionyl chloride should be destroyed by water present in the aqueous sodium hydroxide. The desired amide is soluble in ether, and it may be helpful to add a bit more at this point.
Extraction:Remove the gas trap assembly, the condenser, and the Claisen head. Transfer all the liquid to a separatory funnel. Shake the funnel for about 2-3 minutes. The shaking should help the conversion of any remaining acid chloride to the sodium salt. Allow the layers to separate and then remove then lower aqueous layer, and discard it. Add another 14 mL portion of 10% sodium hydroxide to the remaining ether layer and shake the funnel for
another 2-3 minutes. Drain and discard the aqueous layer once again.
Now add 14 mL of 10% hydrochloric acid, and shake the funnel for
another 2-3 minutes to remove any remaining diethylamine as its hydrochloride salt. Allow the layers to separate, drain and discard the lower aqueous layer.
Transfer the ether layer into a dry erlenmeyer flask and dry the ether phase with granular anhydrous sodium sulfate. Decant the ether phase away from the drying agent into another flask. Evaporate the ether by heating the flask to about 50*C, in a fume hood or with you out of the room. The crude lysergic acid diethylamide will remain.
Crystallization:The freebase should be dissolved in warm, dry methanol (4 mL/ g lsd
TIHKAL#26). This solution should be treated with dry d-tartaric acid (0.232 g/ g LSD
TIHKAL#26). This solution should be treated with EtO
2 under stirring until the solution becomes extremely cloudy, and placed into a light proof container inside a refrigerator to induce crystallization. Further recrystalizations and drying will yeild Lysergic Acid Diethylamine tartrate salt.
This post is dedicated to the late Dr. Timothy LearyHave fun kids!