There was shown an ability of synthesis of indoles by reacting conc. H
2SO
4 with products of reaction of aniline (ArNH
22) with glycidic esters.The main idea was reacting aniline with glycidic esters at high temp (in pipe bomb) on a 1st stage:
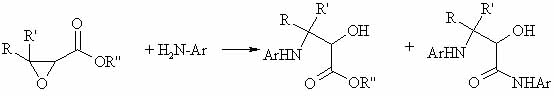
On a 2nd stage
alpha-oxy-
beta-anilinoester (yield 30-84%) is treated with conc. H
2SO
4 at high temp:
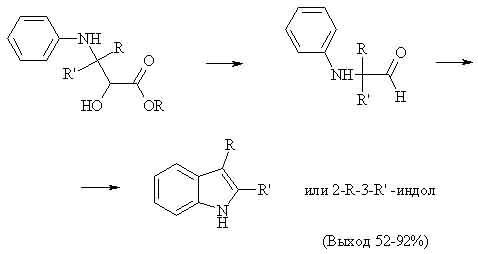 These 2 images are located at geocities,which has traffic limit,you can encounter problem opening them soon:)
These 2 images are located at geocities,which has traffic limit,you can encounter problem opening them soon:)However it was found out that adding conc. HCl to excess of aniline makes opening of epoxy ring more easy, thus making synth of indoles in one stage.
Experimentally reaction is very easy: excess of aromatic amine is mixed with HCl and equimolar amount of glycidic ester,then solution is heated up to 180-190 C,indoles are separated in a common way.The yield of desired products,which have alyfatic or aromatic radicals,is 68-85%.
This was presented by Narayana of HyperLab.
So having corresponding glycidic ester we could get DMT in one stage from aniline.
The main question is could anyone suggest a synth of glycidic esters,for producing exactly (unsubst.)indole???
Patents search gave nill

When I die bury me upside down so the world can kiss my ass.
