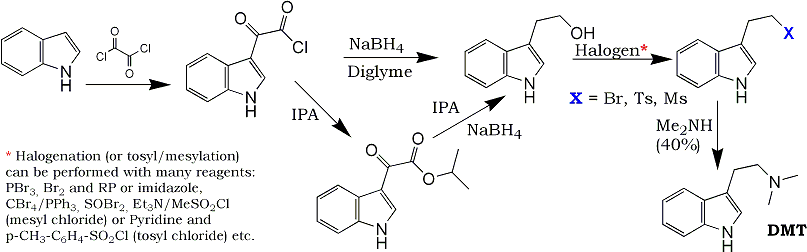
That find was really great! Now we finally have an extremely easy and high-yielding route from Indoles to Tryptophols, which of course are only two steps away from N,N-dialkyltryptamines such as DMT.
First step: Indoleglyoxyl chloride from IndoleThis is performed by adding oxalyl chloride to an etheral solution of indole, and filtering off the precipitated product, using the well-known Speether-Anthony procedure, see
Patent US2825734
for some examples.
Second step (Reduction of the acid chloride directly):As sodium borohydride according to the patent can reduce 3-indoleglyoxylic acid ester and indoleglyoxyl chloride, but not 3-indoleglyoxylic acid itself, care must be taken to exclude all moisture from this reaction, as the acid chloride is hydrolyzed to 3-indoleglyoxylic acid by water. Therefore, this acid chloride is swiftly added to either diglyme (so that it can be directly reduced to tryptophol, as in example 7)
Second step (Reduction of the acid chloride directly):The alternative is to go via an 3-indoleglyoxylic acid ester, in which case the acid chloride is dissolved carefully in dried methanol or isopropanol (heat evolution!) to form the methyl (For Example

or isopropyl 3-indolylglyoxylate (For Example 1), respectively.
As the isopropyl ester is reduced in isopropanol, that method is advantageous, as it becomes a one-pot reaction - when the isopropyl ester has been formed, sodium borohydride is directly added to the same solution. The same cannot be done in the case of the methanol ester, as sodium borohydride decomposes too fast in methanol. It is probably not advantageous to attempt to make the ethyl ester for reduction of this in ethanol, as ethanol almost always contain a few percent water, which as said will hydrolyze the 3-Indoleglyoxyl chloride to the by sodium borohydride irreducible 3-Indoleglyoxylic acid.
After this, go on to brominate (or tosylate) the tryptophol as described earlier in this forum, and aminate the formed bromide or tosylate. Both these steps should work very well.
Patent US4062869
Process for preparation of tryptophols
AbstractA process for preparing tryptophol derivatives comprises reducing a 3-indolylglyoxylic acid ester or acid halide using an alkali metal borohydride in the presence of an alcohol or ether solvent. The tryptophol derivatives prepared are useful as intermediates to pharmacologically active compounds.
This invention relates to a process for the preparation of 1-unsubstituted-3-(2-hydroxyethyl)indole derivatives, such as tryptophol.
Patent US3076814
describes the preparation of 3-(2-hydroxyethyl)indoles by reducing 3-indoleglyoxylic acids using lithium aluminium hydride. The patent also discloses the partial reduction of the 3-indoleglyoxylic acids using sodium borohydride to give 3-indoleglycolicacids; which compounds, it states, can be reduced further to the 3-(2-hydroxyethyl)indoles by lithium aluminium hydride.
It has now surprisingly been found that, in certain solvents, 1-unsubstituted-3-indoleglyoxylic acid esters and acid halides may be reduced with alkali metal borohydride reducing agents to give 1-unsubstituted 3-(2-hydroxyethyl)indole derivatives.
The ability to carry out the reduction to give tryptophols using borohydrides gives the process of this invention advantages over the known process using lithium aluminium hydride. For example alkali metal borohydrides are relatively safe reagents and reduction may be effected smoothly. Reductions using lithium aluminium hydride on the other hand are more hazardous. The present invention, therefore, provides a process which is suitable for large - scale preparations. The use of sodium borohydride is particularly advantageous as this reagent is less expensive than lithium aluminium hydride. Further advantages over the use of LiAlH
4 include the ability to have substituents present on the indole ring which are unaffected by the borohydride reducing conditions and which would be reduced by the more powerful LiAlH
4.
In one aspect therefore this invention provides a process for preparing a 1-unsubstituted-3-(2-hydroxyethyl) indole which comprises reducing a 1-unsubstituted-3-indoleglyoxylic acid ester or acid halide with an alkali metal borohydride in the presence of an alcohol or ether solvent suitable for effecting the reduction.
Many of the compounds of formula I prepared by the process of this invention are useful as starting materials for the preparation of therapeutically active compounds for example, as described in
Patent GB1375836
. The compounds of formula I may also be converted to the 2-(indol-3-yl) ethyl halides using hydroxyl/halogen exchange reagents, e.g. to the bromide using PBr
3 ; which compounds may then be used to prepare therapeutically active compounds as described in
Patent GB1218570
, corresponding to
Patent US3527761
. By "a hydroxyl/halogen exchange reagent" is meant a reagent capable of displacing the hydroxyl group of an alcohol by a halogen atom. Typical examples are PBr
3, PCl
3, sulphonyl chloride, etc.
The 3-indoleglyoxylic acid ester or acid halide derivatives used as starting materials are known compounds or may be prepared by knwon procedures. For example an indole may be reacted with an oxalyl halide to give a 3-indoleglyoxylyl halide which may then be reacted with an alcohol, e.g. an alcohol of formula ROH wherein R is as defined above, preferably in the presence of a base to give the corresponding 3-indoleglyoxylic acid ester. Examples of alcohols of formula ROH are methanol, ethanol, butyl alcohol, benzyl alcohol, m-cresol and the like.
EXAMPLE 1 - 3-(2-Hydroxyethyl)indoleA vigorously-stirred mixture of isopropanol (3 1), methyl 3- indolylglyoxylate (305 g) and sodium borohydride (125 g) was warmed to 40°C - 50°C and held at that temperature until the initial exothermic reaction was complete. The mixture was then heated to reflux for 4 hours, cooled and diluted with water (5 l). After acidification with hydrochloric acid the product was extracted into dichloromethane. The separated extract was washed with sodium carbonate solution to remove 3-indolylacetic acid and then with water. Evaporation of the solvent gave the crude product as a brown oil. This was distilled at 0.5 - 1 mm pressure (vapour temperature 160-190°C ) and crystallised from toluene (1 l) to give the title compound as white to pale brown crystals m.p. 57-59°C. Yield 170 g. (70% of theory).
EXAMPLE 7 - 3-(2-Hydroxyethyl)indoleA suspension of sodium borohydride (22.6 g.) in diglyme (800 ml.) was stirred in a jacketted flask with water cooling. 3-Indolylglyoxylyl chloride (41.5 g.) was cautiously added in small portions then the mixture was heated to 95°C-100°C for 5 hours. The cooled reaction mixture was diluted with water (2 l.), acidified with hydrochloric acid and extracted with dichloroethane. The extract was washed with sodium carbonate solution then with water and evaporated to give crude tryptophol as a brown oil. This was distilled at 0.1 mm and the distillate collected at 155°C-160°C was crystallised from toluene (50 ml.), giving pure tryptophol as white crystals m.p. 57°C-59°C. Yield: 17.7 g.
EXAMPLE 8 - 3-(2-Hydroxyethyl)indoleMethyl 3-indolyglyoxylate (30.4g.) was added to a stirred suspension of sodium borohydride (17.0g.) in ethanol (industrial methylated spirit, 300 ml). the exothermic reaction raising the temperature of the mixture from 20°C to 45°C. After heating to reflux for 5 hours the mixture was cooled, diluted with water (21/2 l.) and extracted with dichloromethane. The extract was concentrated under reduced pressure to an oil, which crystallised from toluene (40 ml.) to give the title compound as a buff-coloured powder, purity 98% by GLC. Yield 15.2g.