Post by: sushitake on September 08, 2003, 06:07:00 PM
Post by: Vitus_Verdegast on September 08, 2003, 06:59:00 PM
I did a quick search on your compound but came up with nothing, except this paper on analgesic tryptamines:
Substituted 4,7-dihydroindoles. Remers, William A.; Weiss, Martin J. (American Cyanamid Co.). U.S. (1969), 4 pp. CODEN: USXXAMPatent US3444174 (http://l2.espacenet.com/dips/viewer?PN=US3444174&CY=gb&LG=en&DB=EPD)
19690513 Patent written in English. Application: US 66-584355 19661005. CAN 72:66809 AN 1970:66809 CAPLUS
Patent Family Information
Patent No. Kind Date Application No. Date
US 3444174 A 19690513 US 1966-584355 19661005
Priority Application
US 1966-584355 19661005
Abstract
Substituted 4,7-dihydroindoles were prepd. from the corresponding indoles by treatment with an alkali metal or alk. earth metal in liq. NH3 in the presence of an alc. For example, a mixt. of 2.4 g 5-methoxytryptamine, 50 ml Et2O, and 500 ml liq. NH3 was treated with 4 g Li wire. After 1 hr, MeOH was added until disappearance of the blue color. Evapn. of NH3 and extn. of the residue gave 1.27 g 4,7-dihydro-5-methoxytryptamine, m. 100-14°. Other 4,7-dihydroindoles prepd. were 4,7-dihydro-2,7-dimethyl-5-methoxy-N,N-dimethyltryptamine, m. 80-3°; 4,7-dihydro-5-methoxy-N,N-dimethyltryptamine, m. 91-3°; 4,7-dihydro-2,7-dimethyl-5-methoxy-N,N-pentamethyl-enetryptamine; 4,7-dihydro-5-methoxyindole, m. 65-8°; 4,7-dihydro-6-methoxyindole; 4,7-dihydro-5-methoxy-1-methyl-N,-N-dimethyltryptamine; 4,7-dihydro-a-ethyltryptamine, m. 76-9°. These compds. have analgesic and central nervous system depressant properties.
Post by: Vitus_Verdegast on September 08, 2003, 07:03:00 PM
Synthesis and analgesic activity of N-(N-acetyl-L-amino-acyl)-5-methoxytryptamines. Dongmei, Han; Chao, Wang; Ming, Zhao; Shiqi, Peng. College of Pharmaceutical Sciences, Beijing Medical University, Beijing, Peop. Rep. China. Preparative Biochemistry & Biotechnology (2000), 30(1), 39-47. CODEN: PBBIF4 ISSN: 1082-6068. Journal written in English. CAN 132:322109 AN 2000:138442 CAPLUS
Abstract
5-Methoxytryptamine was acylated with N-acetyl-L-amino acids to give N-(N-acetyl-L-amino acyl)-5-methoxytryptamines which were evaluated for analgesic by the tail flick test. Among the six compds. that were prepd, the analgesic potencies of N-(N-acetyltryptophanyl)-5-methoxytryptamine and N-(N-acetylglycyl)-5-methoxytryptamine were much more potent than that of melatonin.
Synthesis and analgesic activity of N,N'-dicarbonyltryptamines. Dongmei, Han; Chao, Wang; Ming, Zhao; Shiqi, Peng. College of Pharmaceutical Sciences, Beijing Medical University, Beijing, Peop. Rep. China. Preparative Biochemistry & Biotechnology (2000), 30(3), 231-240. CODEN: PBBIF4 ISSN: 1082-6068. Journal written in English. CAN 133:252248 AN 2000:520139 CAPLUS
Abstract
5-Methoxytryptamine and L-tryptophan Me ester were acylated with malonic acid, di-Me malonate, or succinic anhydride to produce the corresponding N,N'-dicarbonyltryptamine derivs. The analgesic activity was evaluated by the tail flick test. All of the compds. exhibited desirable analgesic potency. This result is consistent with that of N-(N-acetyl-L-tryptophanyl)-5-methoxytryptamine and confirmed that introducing substituted tryptamine into the amide chain of melatonin does enhance analgesic potency.
Post by: Lilienthal on September 09, 2003, 01:22:00 AM
Post by: sushitake on September 09, 2003, 02:33:00 PM
Post by: longimanus on September 10, 2003, 06:19:00 AM
According to Cooper " There has been discovered one compound which may be of minor interest in that it is an analgesic with potency similar to morphine and can be described as a ring condensation product of N,N`,3-trimethyl-5-hydroxytryptamine (Brossi 1985)."
I`ve found this article in Erowid, Rhodium and www.a1b2c3.com and the only difference between the three texts is that in the Erowid variant there is "(compound 46, Figure 14)" in front of "(Brossi 1985)".And since there doesn`t exist N,N`,3-trimethyl-5-hydroxytryptamine I`m dying to see Figure 14.
Post by: sushitake on September 10, 2003, 01:48:00 PM
Post by: Bwiti on September 11, 2003, 12:11:00 AM
Patent US3444174 (http://l2.espacenet.com/dips/viewer?PN=US3444174&CY=gb&LG=en&DB=EPD)
, example 1, I wonder what would be created if something easier to work with, such as diethylamine, is used in place of the ammonia? Maybe it would have analgesic properties?Post by: longimanus on September 11, 2003, 01:10:00 PM
But 1,3-dimethyl-3-(2-methylaminoethyl)-2,3-dihydro-1H-indol-5-ol does.
And Cooper don`t see any difference!?!
I searched Medline for the Brossi`s article and found the following:
Further explorations on unnatural alkaloids
Bossi A.
J Nat Prod 1985 Nov-Dec 48:878-93
Medline (PMID=86142792) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=86142792&dopt=Abstract)
accompanied with the abstract below:
(+/-)-N1-noreseroline-O-methyl-ether, prepared by a modification of the Julian total synthesis, afforded with R-(+)-methylbenzyl-isocyanate two diasteromeric ureas, converted individually into (+)- and(-)-eseroline after thermolysis, N-methylation and O-demethylation. (+)-Physostygmine was obtained after reaction of (+)-eseroline with methylisocyanate. Biological properties of (-)- and(+)-eseroline and (+)-physostigmine will be reported... continuing with info about colchicine and MPTP research
What I know about physostigmine is that it`s an agonist of muscarine and respectively - antagonist of atropine. And then I found the information below:
Physostigmine (eserine),C 15 H 21 N 3 O 2 , MW 275.3, CAS 57-47-6, MI 13: 7469, crystaline solid, soluble in chloroform, toxic - forms a carbamylated enzyme complex with acethylcholinesterase that degrades slowly. Crosses blood-brain barrier.
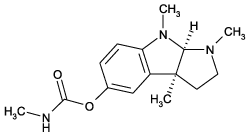
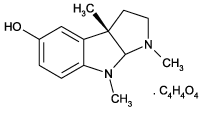
(-)-Eseroline.fumarate, C 13 H 18 N 2 O.C 4 H 4 O 4 , MW 218.3 . 116.1, CAS 104015-29-4, white solid, soluble in water or DMSO, slightly soluble in MeOH, metabolite of physostigmine (eserine), potent analgesic.
References:
Medline (PMID=6293841) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=6293841&dopt=Abstract)
Medline (PMID=7092918) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=7092918&dopt=Abstract)
Medline (PMID=2858526) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=2858526&dopt=Abstract)
So...the analgesic "tryptamine" is actually eseroline. The price of (-)-eseroline fumarate is around 50euro/10mg or 170 euro/50mg and the price of physostigmine is 25euro/100mg (bioresearch grade). Hmm...interesting.
Post by: sushitake on September 11, 2003, 11:09:00 PM
Post by: Lilienthal on September 12, 2003, 12:58:00 AM
Post by: longimanus on September 12, 2003, 06:56:00 AM
Pharmacology:
Medline (PMID=91068238) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=91068238&dopt=Abstract)
it kills brain cells!Medline (PMID=91334549) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=91334549&dopt=Abstract)
Medline (PMID=82231462) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=82231462&dopt=Abstract)
Medline (PMID=89211823) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=89211823&dopt=Abstract)
Medline (PMID=87065740) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=87065740&dopt=Abstract)
Medline (PMID=93209316) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=93209316&dopt=Abstract)
Medline (PMID=81185784) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=81185784&dopt=Abstract)
Medline (PMID=84179927) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=84179927&dopt=Abstract)
Medline (PMID=82014213) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=82014213&dopt=Abstract)
Medline (PMID=98369748) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=98369748&dopt=Abstract)
Medline (PMID=81122677) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=81122677&dopt=Abstract)
Medline (PMID=80074451) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=80074451&dopt=Abstract)
Synthesis:
Medline (PMID=98296296) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=98296296&dopt=Abstract)
Similar to eseroline:
Medline (PMID=92277090) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=92277090&dopt=Abstract)
There is also an article I couldn`t find: `A Formal Synthesis of (+)-Eseroline via an Aza-oxy-Cope Rearrangement` Santos,PF; Almeida,PS; Lobo,AM;Prabhakar,S.; Heterocycles, 2001, 55, 1029-1043.
http://www.cpdd.vcu.edu/DEC/acrobat/DEC1997.pdf (http://www.cpdd.vcu.edu/DEC/acrobat/DEC1997.pdf)
would be ineresting for every opiophile.And, Lilienthal, do you mean the toad, mimosa, etc. 5-MeO-DMT or...cuz I think that "N`" refers to the indole nitrogen?!?
Post by: Lilienthal on September 12, 2003, 08:00:00 AM
Post by: sushitake on September 12, 2003, 11:34:00 AM
Post by: longimanus on September 12, 2003, 12:02:00 PM
"N`" stands for second substituted nitrogen atom. For example N,N`-dimethylehtyleneurea = 1,3-dimethyl-2-imidazolidinone. And which is the second nitrogen in tryptamine? But we are not talking about tryptamine. Look at the structure of eseroline. N,N`,3-trimethyl-5-hydroxytryptamine doesn`t exist. Cooper means 1,3-dimethyl-3-(2-mehtylaminoethyl)-2,3-dihydro-1H-indol-5-ol. As I said - Cooper doesn`t look to be a chemist.
It`s more toxic. Read
Medline (PMID=91068238) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=91068238&dopt=Abstract)
: Eseroline was comparatively more toxic than the parent compound, physostigmine. You can buy eye-drops, hydrolize physostigmine to eseroline and...kill someone slowly. Search for derivates:SAR
Medline (PMID=81122677) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=81122677&dopt=Abstract)
Akuammine
Medline (PMID=92277090) (http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=92277090&dopt=Abstract)
Post by: Lilienthal on September 12, 2003, 01:21:00 PM
Post by: longimanus on September 13, 2003, 01:46:00 AM
http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?rxntypeid=30&prep=CV7P0197 (http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?rxntypeid=30&prep=CV7P0197)
. Do you believe them?Let`s stop this! The theme of the thread is analgesic tryptamine but we`re arguing about nomenclature.
respectfully, longimanus
Post by: xeno_tropic on September 22, 2003, 01:19:00 PM