The proccedure is pretty simple and can be done with fairly good yields.
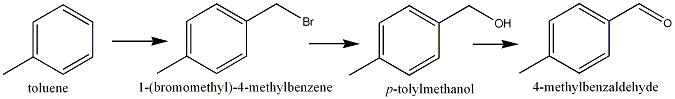
Step 1. Bromomethylation of toluene. This can be done using Paraformaldahyde***, HBr (produced in-stu via NaBr and H2SO4 and GAA as the solvent.) (87% yield)
Reference:
Post 475109
(Lego: "Amphetamines/PEAs w/o benzaldehyde or nitroethane", Novel Discourse)Step 2. Coversion of Toluene to Alcohol (may not be needed). I can't seem to find any data, but I'm sure a base-catalysed hydrolysis would remove the bromine group and leave you with a hydroxyl group. NaBr is regenerated (but NaBr is dirt cheap anyway).
Step 3. Oxidation of Benzyl Alcohol via Oxone, NaBr.* Proceedure uses NaBr, Oxone and CH3CN for the solvent.
I assume that NMP** could be used in place (NMP can be distilled out of several paint strippers) It is a polar solvent. (correct me if I'm wrong) (Yield 96%)
Reference:
https://www.thevespiary.org/rhodium/Rhodium/chemistry/alcohol2aldehyde.oxone-nabr.html
The advantages are that this proceedure is cheap, much cheaper than buying the tolualdahyde. And toluene ain't that hard to come by. 1 Gallon = $10 dollars?
Does anyone have a reference proceedure for Step 2. I know it should be a relatively easy proceedure.
* There are many other ways to do this Oxidation, DMSO and catalytic HBr could be used. In which case the hydrolysis (Step 2) becomes uncess. It seems though, that yields are higher when oxidation is done w/ the alcohol.
** NMP is found in a 70-85% concentration in some paint strippers.
*** The paraformaldahyde can be replaced by trioxane which are sold as fuel bars.
Post 442563
(Rhodium: "Trioxane as (para)formaldehyde substitute", Methods Discourse)90g for ~$0.95. Keep in mind the bars must be cleaned. The outside layer needs to be scaped off, and the rest of the bar crushed up and washed with acetone to remove the color.