Aromatic Allylation via Diazotization: Metal-Free C-C Bond FormationF. Ek, O. Axelsson, L-G. Wistrand, T. FrejdJ. Org. Chem. 67(18), 6376-6381 (2002)
(
https://www.thevespiary.org/rhodium/Rhodium/pdf/allylation.diazotization.pdf)
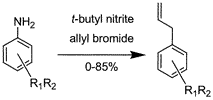 Abstract
AbstractA new method for the synthesis of allyl aromatic compounds not involving any metal-containing reagent or catalyst has been developed. Arylamines substituted with a large number of different substituents were converted via diazotizative deamination with tert-butyl nitrite in allyl bromide and acetonitrile to the corresponding allyl aromatic compounds. The allylation reaction was found to be suitable for larger scale synthesis due to short reaction times, a nonextractive workup, and robustness toward moisture, air, and type of solvent.
DOI:
10.1021/jo0258103
Synthesis of Allyl-3,5-dinitrobenzeneNeat 3,5-dinitroaniline (75 g, 0.41 mol) was added in portions to a solution of tert-butyl nitrite (84.6 mL, 0.71 mol) and allyl bromide (530 mL, 6.15 mol) in CH
3CN (25 mL), keeping the temperature between 11-15°C. Before the addition of the final 25% of 3,5-dinitroaniline, more tert-butyl nitrite (21 mL, 0.18 mol) was added. The reaction mixture was then stirred at room temperature (23-25°C) for 1 h. Excess tert-butyl nitrite, allyl bromide, and CH
3CN were distilled off from the reaction mixture at reduced pressure, and toluene (500 mL) was added to the orange-brown residue.
The resulting mixture was filtered twice through alumina pads (10×10 cm), and the pads were washed with a total of 1.5 L of toluene. The toluene was distilled off at reduced pressure, and isooctane (200 mL) was added to the remaining pale yellow residue. The mixture was stirred at 60°C for 0.5 h (to extract the partly polymerized allyl bromide and 1,2,3-tribromopropane from the product) and then cooled to approximately -50°C. As soon as a white precipitate (partly polymerized allyl bromide and 1,2,3-tribromopropane) started to form, the solvent was decanted (including the precipitate) and to the remaining yellow oil another portion of isooctane (200 mL, 20°C) was added. The oily residue in isooctane was then stirred at -50°C with a spatula until crystals formed. In some cases, it was necessary to decant the isooctane phase and add fresh isooctane before the oil started to crystallize. The crystals (remaining in the flask) were then washed twice with isooctane (precooled to -50°C) at -50°C. Residual isooctane was removed at reduced pressure to give 78.9 g of Allyl-3,5-dinitrobenzene as a yellow oil containing less than 4% of 3,5-dinitrobromobenzene according to
1H NMR analysis. This corresponds to 89% yield of allyl-3,5-dinitrobenzene. In an alternative procedure, the crystals (after washing with isooctane at -50°C) were collected by filtration at -50°C and then washed once with isooctane (precooled to -50°C). The latter method gave the same yield of slightly purer Allyl-3,5-dinitrobenzene.
I believe that this workup is definite overkill, unless dealing with a very expensive substrate, or if the substrate forms a large amount of by-products. I'd suggest that after the removal of the reaction solvents and excess allylbromide (recover, purify and re-use), the crude product is diluted with an equal amount of diethyl ether (or possibly pet. ether), then washed twice with 5% HCl, twice with 10% aqueous NaHCO3 and once with saturated aqueous NaCl, then dried over anhydrous MgSO4, filtered and the filter cake washed with a little ether. The solvent is then distilled off (recover, purify and re-use), and the residue is then fractionately distilled (under vacuum, if necessary) to isolate the pure allylbenzene.