From ChemExper (
http://www.chemexper.com/
):
N-selectride
Synonyms: -
Molecular Formula: C
12H
28BNa
Formula Weight: 206.15
Registry number: 67276-04-4
From Sigma-Aldrich search under name:
(
http://www.sigma-aldrich.com/sacatalog.nsf/Prod?OpenFrameSet&%2A=/sacatalog.nsf/ProductLookup/Aldrich213403?OpenDocument%5E
)
213403 N-Selectride®
(sodium tri-sec-butylborohydride, 1.0M solution in tetrahydrofuran) , 67276-04-4
Synonyms: (sodium tri-sec-butylborohydride, 1.0M solution in tetrahydrofuran)
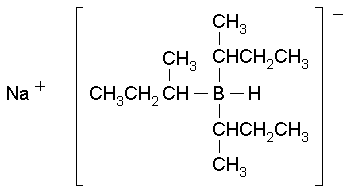
Molecular Formula: NaB[CH(CH
3)C
2H
5]
3H
Molecular Weight: 206.16
CAS: 67276-04-4
MDL Number: MFCD00011709
Density: 0.893
Comments: Flash Point (°F): 5
Product Comments: (Packaged under nitrogen in Sure/SealTM bottles)
®Registered trademark of Sigma-Aldrich Co.
ELINCS/EINECS Number: 999-999-9
Miscellaneous: Label Precautions: Flammable liquid
Corrosive
Reacts violently with water
Harmful liquid
Handle and store under nitrogen
May develop pressure
From Certificate of Analysis on same page:
APPEARANCE : CLEAR COLORLESS LIQUID
HYDROGEN EVOLUTION : 1.01 M
QUALITY CONTROL ACCEPTANCE DATE : JULY; 2001
More technical details:
javascript:returntostart('/aldrich/bulletin/al_techbull_al123.pdf')
Quantitative Analysis of Active Metals and Metal Hydrides via Gas Buret.
You find there also that Sodium borohydride, 0.5M in diglyme ( 2-methoxyethyl ether ) and Sodium borohydride, 2.0M in triglyme ( triethylene glycol dimethyl ether ) are the latests solutes used for storage and use of borohydrides. (Hint,hint,hint, Aa, when does
someone see the light?)
From Structure Image:
http://www.sigma-aldrich.com/sacatalog.nsf/productlookup/Fluka71320?OpenDocument
Literature References: Reviews: H.C. Brown, S. Krishnamurthy, Tetrahedron 35, 567 (1979); G.W. Gribble, C.F. Nutaitis, Org. Prep. Proc. Int. 17, 317 (1985); Reduction of carboxylic acid derivatives: S.R. Wann, et al., J. Org. Chem. 46, 2579 (1981); E. Santaniello, et al., ibid. 46, 4584 (1981); K. Soai, et al., Synth. Commun. 12, 463 (1982); A. Giannis, K. Sandhoff, Angew. Chem. 101, 220 (1989); Reduction of acid chlorides to aldehydes: J.H. Babler, Synth. Commun. 12, 839 (1982)
And that long post of mine about that fabulous website of that US professor about imines, you have to look that one up again.
http://www.sigma-aldrich.com/aldrich/bulletin/al_techbull_al123.pdf
(was not the right link up there with that java script)
Here come a few very interesting ones:
http://www.sigma-aldrich.com/al_organics/pdfs/naa31_1.pdf
Organic Syntheses via Boranes, Volume I and II, prof. H.C.Brown from Purdue Uni.
Synthetic Applications of Zinc Borohydride:
http://www.sigma-aldrich.com/al_organics/pdfs/rv2_31_1.pdf
Nice piece of research, look at the references, don't forget to read
nr.2, and all the ones with those funky Indian names in them. He worked 4 years with prof.H.C. Brown at Purdue University.
And then they went on in India to research further, and came up with that link I gave once in Serious Chemistry as an answer to RevDrone's question about the inner workings of the NaBH4 onepot.
(Zinc borohydride has unique properties. Unfortunately, it is also not stable long enough to be offered commercially; hence,
it must be prepared in situ.)
http://www.sigma-aldrich.com/al_organics/pdfs/mbah31_1.pdf
( METAL BOROHYDRIDES )
Read the references from Dr. Narasimhan.
Ehh, read this one too:
http://www.sigma-aldrich.com/al_organics/pdfs/iso31_1.pdf
Magtrieve superoxidant ( CrO2 ).
Ehh, threw this one also in, can't resist:
Aldrich Techware LaBNoteBook, lots of interesting toys:
http://www.sigma-aldrich.com/al_organics/pdfs/ins31_1.pdf
and then look at this baby:
CORNING MODEL 611 HIGH VOLUME STIRRER
Newly designed to stir high volumes of
liquid (up to 5 gallons) with ease.
• One year warranty
• 120 volt; 100 to 1,100rpm stir range
• Rugged ABS top, 11 x 11 inch.
WISDOMwillWIN