Apparatus and method for the production of anhydrous stannic chloride and a questionApparatus: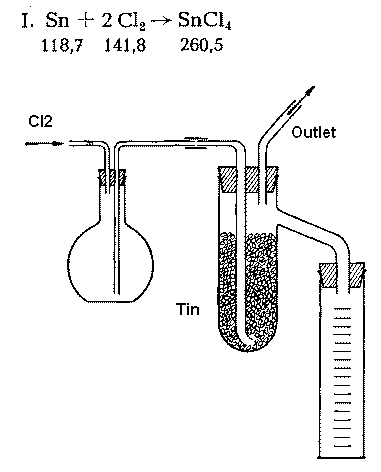
from: Georg Brauer: Handbuch der präparativen anorganischen Chemie, 3. Ausgabe, 1975
Additional information:
"Method of making anhydrous tin chlorid, 1912, to Castner Electrolytic Alkali Company"
Patent US1018805
To the picture: The chlorine reacts with the granulated tin and the increase of volume drives the SnCl4 which is a liquid over into the collecting flask. If some SnCl4 is by hand it is to be added to the tin - this guarantees a smooth start of the reaction. The reaction vessel is first to be heated and after the reaction kicked in to be cooled by a waterbath.
If no SnCl4 is used at start higher temperatures are needed to get it going. In this case it is favorable to adapt an reflux condensor instead of the outlet tube for the reaction will get hot fast and one doesnt want to loose the SnCl4.
Outlet tube or condensor are to be connected to a fumehood or a caustic scrubber - the outlet ending not IN but directly OVER the surface of the NaOH solution.
To the patent:The patent tells us that it is not necessary to use absolute dry chlorine and pure tin if the SnCl4 is distilled lateron. Hydrated SnCl4 drops out as crystals anyways - a nice feature of tin

Anybody not so experienced in chemistry and anybody not owning a fumehood is strictly advised to use the suggested reflux condensor design or he/she/it will regret it badly. Promised. I did.
QuestionWill Al added to anhydrous SnCl4 reduce this to the metal or to SnCl2?
Or - gasp! - not at all?
Steel is inert to anhydrous SnCl4.
I guess it should be the metal, but whats sure in chemistry?