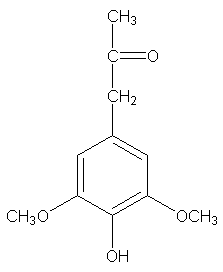
Seen any interesting molecules around lately? This is one of the Hibbert ketones available from wood lignin. I'll call it syringyl-2-propanone, just as a friendly name. Seems like some beez might show enough interest in this, to wonder where to get it. You might say the woods are full of it, it grows on trees. This is another aspect of the lignin story;
Post 190600 (missing)
(halfapint: "Lignin Syringaldehyde Again", Novel Discourse).
Hibbert ketones are derived from lignin by a different route, not by the metal salt oxidation in alkali by which syringaldehyde and vanillin are obtained. Most often, Hibbert ketones are produced by alcoholysis with HCl, but that's not the way we should use, as I'll show you. The Hibbert ketone which corresponds to vanillin, I'll call vanillyl-2-propanone.
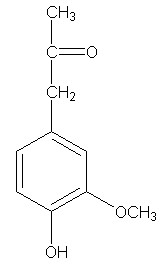
Now some folks around here might also see some good possibilities in this molecule. As foxy2 just showed us, on a LabTop scale no less, if you
first alkylate the 4-hydroxy, and
then brominate, the bromine will go in the 2-pocket, not the 5-hole. Sodium methoxide treatment will methylate the 2-bromo compound, leaving you just a reductive amination away from a real good time. There's got to bee a catch: oh, yeah. There's a catch all right. You have some more Hibbert ketones that look like vanillin, in their ring substitution pattern:
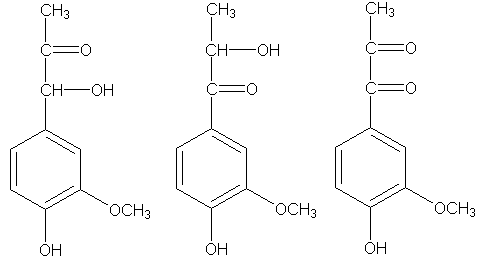
Well, if that's not bad enough, you also have some more, which have their rings substituted like
syringaldehyde:

!!!
Ready to give up yet? I was going to tell you why not to use ethanolic HCl to chop lignin into Hibbert ketones. The reason, is that if you use alcohol, you get a Hibbert ketone that has the vanillin ring pattern, with oxygen on the propyl chain's 1 position, and an ethyl
ether on the 2 position! You can't bust up that ether without also losing your phenol ether, a giant step backward. Useless. It's got a sibling, with the ethyl ether on the 1 slot, and the ketone in the 2 propyl position. Need I say, there are also syringyl versions of both of these? That makes four useless compounds, out of a round dozen Hibbert ketones! Horrible. So we don't use alcohol, we use dioxane.
Dioxane, by itself, well with a bit of water, will digest the lignin out of wood. But I would suggest not using wood. Use
cardboard boxes, known as corrugated paper cartons on Sunday.
I have been able to get more lignin out of cardboard boxes than from wood. I don't know why. Perhaps extra lignin is added as a "sizing", whatever that is. Perhaps it's a stiffening agent, or added for strength. Or could be, it's just concentrated in the production process. That's the key to your future success: you're in the recycling business now. We're talking about changing
over ten per cent by weight of cardboard into precursors for psychedelic drugs. Antoncho, are our tanks in Berlin yet? As pOpEye said, "A drug war would be FUN!"
So after we extract the lignin with dioxane, we turn essentially all of it into Hibbert ketones by adding dilute hydrochloric acid to the dioxane. (My dioxane was cheap, for I distilled it out of ethylene glycol, actually antifreeze, with dilute sulfuric acid. It's wet; so?) I know, you're still dubious about our eight Hibbert ketones, how we are ever going to separate them. We can get them away from everything else by neutralizing the acid, condensing the solvents, then precipitating their condensation products with saturated sodium bisulfite, but how are we going to separate the 2 good ones from the waste? There won't be any waste, we're going to use them all.
To start with, the vanillyl-2-propanone is the lightest molecule there. In a fractional vacuum distillation, it will be the lowest boiling ketone, and it will come over first. One down, seven to go. The next three vanillin-like compounds can be combined. Then the syringaldehyde-like compounds will start to come over. The syringyl-2-propanone will again be the first of that lot. It is the most nearly finished of all these compounds, for it only needs 4-alkylation and reductive amination before it can go out the door. After that, the other syringyl compounds can also be combined, for it doesn't matter to us which ones they are. So we collect 4 fractions, in 4 bottles: vanillyl-2-propanone, the vanillyl keto-alcohols and diones (diketones), syringyl-2-propanone, and the other 3 syringyl ketones.
The mixed ketones we reduce to diols. Know what to do with vicinal diols? Of course, convert them all to 2-propanones, by treatment with lithium iodide or sulfuric acid! Then you can merge
all the lignin aromatics into just 2 bottles, vanillyl-2-propanone and syringyl-2-propanone! That is how you can convert more than 10% of the weight of cardboard, into precursors.
So have I done this? No, I just now figured it out. I am easily excited, and I wanted to share this feeling with you while it is fresh. Plainly, I could be setting myself up, because I do not know the boiling points of these compounds. If the boiling points of the vanillyl and syringyl groups overlap, it would mess up my proposed separation. I also do not know the relative abundances of these ketones. This would vary with lignins.
The salient point is the reduction of the mixed ketones to diols, removing their differences by making them identical compounds. If the syringyl-2-propanone cannot be separated out by distillation, it will be reduced into the 2-propanol and probably wasted, when all the other syringyl compounds are converted into what it used to be: syringyl-2-propanone.
turning
science fact into
<<science fiction>>