WOWWWWWWW!!!!!!!!!!!Un-fucking-believable!!!
I think i just acquired another 'goal of my life'!
Sorry if i sound like an underscrewed science geek

- for i am not. But this is just SOOOOOOOOOOOOOOO awesome!
I mean, i always thought that making sexual stimulants like Viagra was far out-of-reach for an amateur chemist, and now it turns out it's fucking POSSIBLE - and, moreover, it's a
phenethylamine!
Just let me quote some passages.
According to the present invention there are provided pharmaceutical and veterinary compositions for use as sex stimulants in male and female mammals comprising as active ingredient a compound of the general formula where X designates --OCH3.--SCH3 or a halogen, the substituent being either in ortho or in meta position, as well as the salts and complexes of these. The term halogen in this context designates chlorine or bromine ... The ortho-chloro, meta-chloro, ortho-bromo and meta-bromo compounds were tested on animals and all of these were found to be effective sex stimulants.
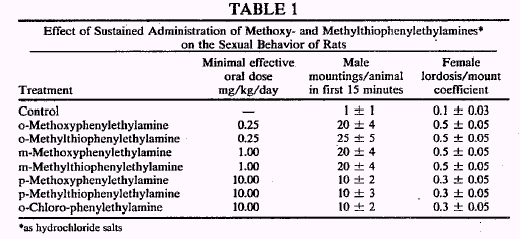
Preliminary tests with human volunteers have shown that the pharmaceutical compositions of the present invention exert a pronounced effect on human males. Tests have shown that o-methoxy- and o-methylthiophenylethylamine are effective in dosages of 35 to 50 mg when given per os as the hydrochloride salt. A preferred dose is in the range of from 5 to 10 mg, a number of times per day. The drug is advantageously administered per os in the form of capsules. The effect is obtained after a few hours to a few days.
So, the only downside (as compared to sildanefil) is the relatively long latency period, but WTF (20x increase in sexual activity is well worth a few hrs wait, IMHO 



 ).
).
Another question - can someone explain me what they meant by 'female lordosis/mount coefficient'? I.e., is there a chance that it could have a similar activity for ladies? That'd bee really cool 
Now, to the synthetic ideas.
Ortho-MeO-PEA seems to bee the most potent of them all. To make the aldehyde which would bee converted by standard means into the PEA, one could:
a) Nitrate toluene (a la Cheapskate).
b) Reduce to toluaniline (again a la Cheapskate, but
Patent US3798271
has this rxn in its most perfect form , yield circa quantitative) and steam-distill the ortho-isomer out.
c) Diazotize/methanolize the amino group (or Sandmeyer it to smth else, if desired)
d) Oxidize the methyl group to aldehyde by any known way, e.g. w/Na peroxodisulfate/FeSO4; or chromic anhydride/PTC
Alternatively, one could try to make the aminobenzaldehyde directly from nitrotoluene, similarly to
http://orgsyn.org/orgsyn/prep.asp?prep=cv4p0031
- there they make para-amino-BA from p-nitrotoluene w/Na2S; can anyone suggest if the same route would bee applicable to ortho-isomer? Seems likely.
Another Q: how does one separate o- and p- nitrotoluenes resulting after intration? Will simple fractional distillation do?
Does anyone have better/shorter ideas? Of course, o-MeO-BA could bee easily bought from a chem supply company. Moreover, ortho-cresol, which is also widely available and cheap (not for SWIM, but anyway) would serve as an excellent precursor.
Dr. Heckyll, my HUGE thanks to you!Antoncho
P.S. Another idea......that has just occurred to me is actually MUCH simpler:
a) Brominate plain benzaldehyde
b) Methoxylate the haloid (the rxn will run much more smoothly than in case of vanillin beecause of the absense of electron-donating substituents on the ring) or, alternatively, hydroxylate/methylate it.
Voila! - here's your meta-MeO-benzaldehyde!