Hello,
My small (worthless??) contribute to the hive. I am writing this because the hive as given me so much. Right now SWIM is out of the country and away from his lab so he wont be able to experiment for a little over a month. When he comes back this is what SWIM plans to do...
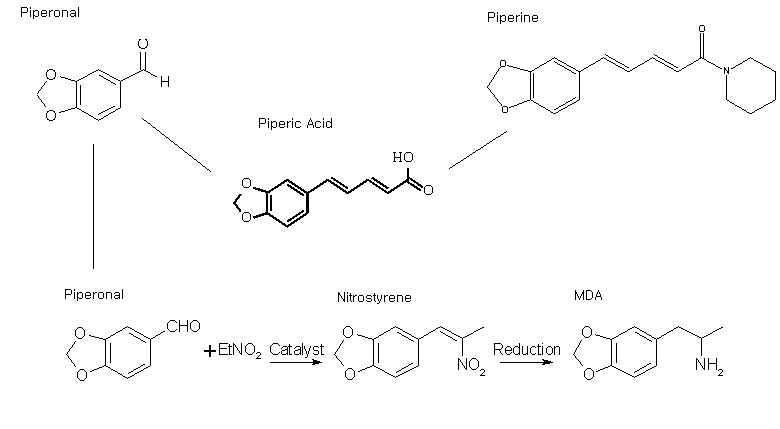
SWIM is going to experiment with all his chemistry knowledge and abilites to synthesize MDA from pepper. SWIM knows it can be done, with hard work and lots of experimentation, but since the sources of sassafras oil is diminishing, this has forced myself and other bees to look into other alternatives.
So when SWIM gets back SWIM is going to go to the grocery store to buy a few pounds of black pepper to extract piperine. SWIM would like other bees to suggest and maybe add information for helping him do this project.. It might take a few weeks but it will get done. SWIM is determined to finish it and make MDA from this. This is what SWIM is thinking..
SWIM has gathered sources and he estimates that the average amount of piperine that black pepper has is 5%. So when SWIM buys about 2KG of black pepper he estimates he might get around 90g piperine if his extraction techniques are good. This is what swim understands...
Pepper ---
EtOH, MeOH, or CH2CL2--->
piperine---
Refluxed with 10% alcholic KOH--->
potassium piperinate----
HCl--->
piperic acid----
KMnO4-->
piperonal SWIM is sure there are other things that can change piperic acid into piperonal. SWIM is willing to undergo most experiments people suggest if they are in his abilites. some other information SWIM has found for the chemicals made in this process....
piperine From MERCK...
Molecular formula: C17H19NO3
Molecular weight: 285.34
Properties: Monoclinic prisms from alcohol, mp 130C. Tasteless at first, but burning
aftertaste. Neutral to litmus. pK (18C: 12.22. Almost insol in water (40 mg/liter at
18C, in petr ether. One gram dissolves in 15 ml alcohol, 1.7 ml chloroform,
36 ml ether. Sol in benzene, acetic acid.
Melting Point: 130C
piperidine hydrochlorideDerivative: Hydrochloride,
Molecular Formula: C5H11N.HCl,
Properties: orthorhombic prisms from alcohol, mp 247C.
Freely sol in water, alcohol
Melting Point: 247C
piperic acid Molecular formula: C12H10O4
Molecular weight: 218.21
Properties: Nee dles from alc. Colorless when freshly prepd, rapidly turns
yellow on exposure to light. mp 216-217C. Sublimes as yellow needles with
partial decompn. uv max (methanol): 340 nm (epsi 28800) . Sol in 50 parts
boiling alcohol, 275 parts abs alcohol at 25C Practically insol in water,
ether, benzene, carbon disulfide.
Melting Point: 216-217C
piperonal Molecular formula: C8H6O3;
Molecular weight: 150.13.
Properties: Colorless, lustrous crystals, mp 37C. Heliotrope odor. bp ~263C;
bp(0.5) 88C Sol in 500 parts water; freely sol in alcohol, ether. Keep in
cool place protected from light. LD50 orally in rats: 2700 mg/kg (Hagan).
Boiling point: bp ~263C bp(0.5)88C
Melting Point: 37C
Piperonylic AcidMolecular formula: C8H6O4 ;
Molecular weight: 166.13.
Properties: Prisms (by sublimation), needles from alc, feathery crys tals
from water. mp 229C. Sublimes around 210? Slightly sol in
water, chloroform, cold alcohol, ether. Absorption spectrum: Dobbie,
Lauder, J. Chem. Soc. 83, 621 (1903)
Melting Point: 229C
MDA Freebase molecular weight= 178.7g/mol
MDA.HCL molecular weight= 215g/mol
Information from Rhodiums site.... Once SWIM gets this PIPERONAL SWIM Plans to use the nitrostyrene method.
Basically piperonal is used as a starting material and condensed with nitroethane to make a nitrostyrene compound which then can later be reduced to MDA.
From "The Complete Book of Ecstasy"
Method 1: To a solution of 15 grams of piperonal in 80 ml glacial acetic acid, add 15 ml nitroethane followed by 10g of cyclohexylamine. The mixture is heated with a boiling water bath for 6 hours. Remove from heat, dilute with 10ml water and cool in an ice bath overnight (place ice bath and reaction mixture in the refrigerator to keep the ice from melting too quick). Bright yellow crystals should precipitate. These are filtered from the cold solution and allowed to dry. Yield ~10g.
A general procedure described by Gairaud and Lappin[45] uses ammonium acetate as the base catalyst for the condensation. It is as follows:
Method 2: 5g of piperonal, 5ml of nitroethane ans 2g of ammonium acetate are added to 20 ml of glacial acetic acid. The resulting solution is refluxed for two hours and then poured into ice-water. The solution is cooled in an ice-bath overnight and the crystals filtered out and allowed to dry. Reduction is the tricky part. There are several ways to reduce the nitrostyrene material into MDA.
*Lithium Aluminum Hydride (maybe easy)
*electrochemical cell (i have some experience with these) 
*possibilty to reduction using metals such as zinc powder and acetic acid, tin powder and hydrochloric acid, or iron powder and hydrochloric acid may work. (downside might not work)The general method of using these reagents is covered in Robert L. Augustine, Reduction: Techniques and Application in Organic Synthesis under the chapter Dissolving Metal Reductions.
SWIM plans to experiment with all those methods to see which one works and which one can give me the best yields..
Nitrostyrene preperation uses piperonal, nitroethane, and a catalyst for the condensation. Some catalysts are cyclohexylamine (hard to get??) or ammonium acetate.For the reduction of the nitrostyrene to MDA the LAH seems to be a good option as it is more experimented with than metal and acid. LAH method needs THF(tetrahydrofuran)(substitute maybe???) so that might be a little tricky to get... There is not much information on reduction so SWIMS going to have to tediously do his own experimentation (any good tips on this???) After reduction is complete, extract the oils, do A/B seperation, do some washings, and probably want to distill the MDA freebase then crystalize

.
Reduction of nitrostyrene with electrochemical cell seems easy enough....
copied from "The Complete Book Of Ecstacy"..
The cells are basically a container divided by some material which will allow charged ions to pass and therefore allow electricity to flow. The membrane also halts the flow (or reduces it considerably) of the material being reduced. This is necessary because if the membrane wasn't there, the reduced product could travel to the other electrode and become oxidized. The membranes are made of such materials as sintered glass, cellophane, porous ceramic plates etc.
This procedure was written for the analogous nitrostyrene which would lead to amphetamine. It should work equally well for MDA[46].
207g (1 mole) of the nitrostyrene is dissolved with a solvent prepared by mixing one liter of ethanol with 500ml of acetic acid and 500ml of 12N sulfuric acid. The solution is placed in the cathode compartment of a divided electrolytic cell containing a mercury, copper or metal of similar nature as the cathode electrode. The anode can be made of lead. 3N sulfuric acid is placed in the anode compartment. Current is passed equaling ~0.2 amperes/cm2 of cathode surface. The temperature is kept between 30-40°C during the electrolysis until at least 8 Faradays of electricity have passed through the solution.
The number of square centimeters of your cathode surface (count both sides if both sides are in contact with the solution) times 0.2 is the amount of current you need flowing through your cell. 1 Faraday is equal to 96485.309 coulombs/mole of electrons and 1 Ampere is equal to 1 coulomb/second. The amount of time in hours the reaction should be run is then 771882.5 divided by the number of Amperes divided by 3600. If you have a total cathode electrode area of 40 cm2 then you need 8 Amperes running through your cell for 26.8 hours.
Remove the ethanol and ethyl acetate present through distillation (quit distilling when the temperature approaches 100C). Basify the remaining solution with 25% NaOH and extract the MDA from the solution with 3 portions of ether (or toluene or methylene chloride). Wash the extract with several portions of calcium carbonate solution, water and then dry with magnesium sulfate. Filter out the drying agent. Pass HCl gas through the solution until no more precipitate is formed. Filter the crystals and wash with ether and allow to dry. There you have it complete hypothesis from piperonal to MDA.HCLOh yeah, also SWIM will try to give much information as possible when doing these experiments. SWIM can distill, fractionally distill, reflux, and do tritrations and just the basic stuff but SWIM cant do TLC analysis or any of that advanced organic chemistry stuff. SWIM wants to help out the hive as much as possible because the ability to get certain precursors are diminishing. SWIMS dream is if this synth/experimenting is successful, I will write a detailed write-up and everyone will be making mda from pepper

hehehe

. What can they do now, put pepper as a List I precursor?? LoL
If possible and if interested other bees can join SWIM and combine data so future bees can easily make MDA from pepperMore detailed information can be obtained at Rhodiums site if interested and more information needed.
 In my opinion MDA is just as great as MDMA so this might be worth it to make for me at least
In my opinion MDA is just as great as MDMA so this might be worth it to make for me at least 