Well, it could be Chromyl chloride (CrO2Cl2): Both are blood-red liquids at room temperature and have similar irritating odors. Chromyl chloride, however, is less dense (d. 1.911) than bromine (d. 2.928) and also more transparent to light.
Chromyl chloride belongs to the class of compounds known as acid chlorides, in which chlorine has replaced the ionic HO groups of the corresponding acid, in the present instance, chromic acid (H2CrO4, Figure 1):
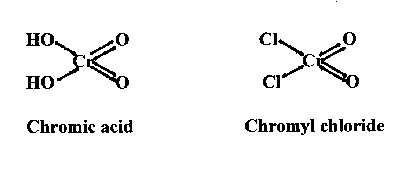
As can be seen, chromium in these compounds is in its hexavalent state with oxidation number +6.
Chromic acid, and the related Dichromic acid (H2Cr2O7) and Chromium trioxide (CrO3) are strong oxidizing agents. If sodium chloride is heated with potassium dichromate and dilute sulfuric acid, the chloride will be oxidized to chlorine:
(1) 2Cr+6 + 6Cl- -> 2Cr+3 + 3Cl2However if these reagents are heated with concentrated sulfuric acid a displacement reaction occurs with formation of the acid chloride:
(2) H2Cr2O7 + 4HCl -> 2CrO2Cl2 + 3H2OThe oxidation state of the chromium remains unchanged in this reaction, and so chromyl chloride, like chromic acid, is a strong oxidant.
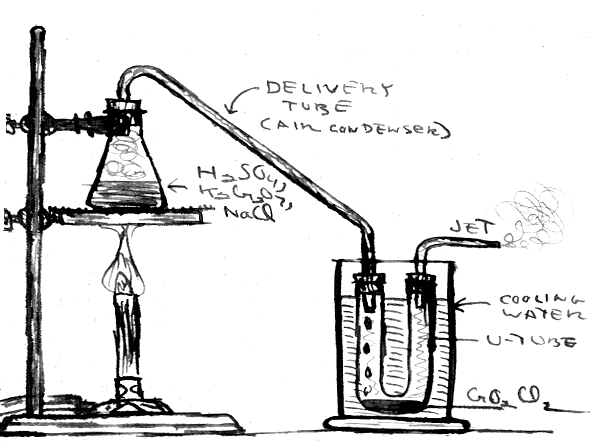 Figure 2.
Figure 2. It is not a difficult matter to prepare small quantities of this reactive liquid in the home laboratory. The chromyl chloride (b.p. 117 C) is recovered by distillation from the reaction mixture. Since the vapor and liquid attack organic materials (including rubber stoppers) it is preferable to work with all glass apparatus. If you're fortunate enough to own or have access to a good selection of standard taper ground joint ware, or that venerable symbol of the chemist, a glass retort, then you'r e all set. Otherwise, don't despair; I used the apparatus shown in Figure 2 with satisfactory results. Rubber stoppers were used; these may have to be replaced after one or two uses in the corrosive environment. Applying a thin coating of silicone stopcock grease may retard attack The air condenser was bent from a length of glass tubing. I recommend neon sign tubing, which is easily worked in a gas flame. If you have some glass working expertise, you can blow a bulb in the riser to serve as a trap for any liquid that may spurt over. The condensed CrO2Cl2 is collected in a U-tube cooled by immersion in a jar of cold water.
The distillation should be carried out in an efficient fume hood or in a well-ventilated area, preferably open air, as CrO2Cl2 vapor is toxic and extremely irritating(1).
Thoroughly mix 20 g potassium dichromate and 8 g sodium chloride (don' t use iodized table salt) and place in a 500 mL Erlenmeyer flask. Add 100 mL conc. sulfuric acid and attach the condenser and receiver. Heat cautiously with a Bunsen flame, taking care that the mixture doesn't froth and spurt over into the condenser. Continue distillation until no more material distills over. Theoretically, the yield should be about 20 g (10 mL) of CrO2Cl2. Store in a glass-stoppered amber glass bottle. Preferably it should not be stored for a long period of time (months or years) as the vapor tends to penetrate the ground joint and freeze the stopper into place. Also the possibility of its falling into the hands of folks unacquainted with its hazards should not be dismissed. To dispose of it safely, pour the liquid slowly into a large volume of cold water, observing ordinary safety precautions (adequate ventilation, protective clothing and eyeware). The resulting dilute solution of chromic and hydrochloric acids can go down the drain as-is, or after neutralizing the acids with baking soda.
Chromyl chloride is a very reactive liquid and fumes strongly in air with evolution of fumes of chlorine and hydrogen chloride. On one occasion when I unstoppered a bottle of CrO2Cl2 the liquid appeared to boil violently, presumably from evolution of these dissolved gases. Initially chromyl chloride is hydrolyzed by moisture:
(3) CrO2Cl2 + 2H2O -> H2CrO4 + 2HClThis is followed by the redox reaction,
(4) 2H2CrO4 + 6HCl -> Cr2O3 + 3Cl2 + 5H2OFor electrochemical balance two equivalents of Cr are reduced from the +6 to the +3 oxidation state and six equivalents of chlorine are oxidized from the -1 to the 0 state.
To illustrate the energetic oxidizing power of chromyl chloride, place a small pool of alcohol (methanol, ethanol or 2-propanol will do) on a watch glass and allow a drop of CrO2Cl2 to fall onto it The alcohol will ignite and burn.
Place a mL or two of CrO2Cl2 in a test tube supported in a test tube rack. Cautiously add a little powdered sulfur from a spatula. The mixture reacts violently, accompanied by a crackling sound and flashes of light and the tube erupts, flinging out excess sulfur and a violet-colored ash.
Similarly, a bit of white phosphorus burned on contact with CrO2Cl2 , though not with the violence encountered with finely divided sulfur.
Other combinations can be tried by the amateur. Powdered metals (e.g., Al, Mg, Zn) should react. It's been reported than ammonia ignites and burns on contact with CrO2Cl2. ; (You can generate a stream of ammonia (NH3) by letting conc. ammonium hydroxide drip on dry sodium hydroxide in a gas generating flask.)
Chromyl chloride is used in organic synthesis of aldehydes and ketones (Etard reaction) and in the preparation of various coordination compounds of chromium.
Notes:1. Treat this compound with respect! Severe burning and blistering occurs if it gets in contact with skin, with severe or prolonged contact leading to ulceration. Moist tissues such as the nasal mucosa and respiratory tract are particularly susceptible to damage, hence the need for adequate ventilation of the work area.
Source:
http://www.sas.org/E-Bulletin/2002-04-19/chem/column.html