3-Prop-2'-ynylindoleW. R. N. WilliamsonJ. Chem. Soc. 2834 (1962)Brown, Henbest, and Jones
1 reported their failure to prepare 3-prop-2'-ynylindole by treating 3-indolylmagnesium bromide with prop-2-ynyl bromide,
"chiefly because the Grignard reagent reacted first with the active acetylenic hydrogen atom to regenerate indole". They do not report the solvent used, but it was presumably diethyl ether. According to Majima and Kotake
2 anisole is a superior solvent for the preparation and subsequent reaction with carbonyl compounds of indolyl Grignard reagents. This apparently cannot be attributed to the higher boiling point of anisole, since in this solvent ethylmagnesium iodide evolves ethane on treatment with indole in the cold, while in ether no gas is evolved at room temperature and heat is required. When we used anisole in the preparation of 3-indolylmagnesium iodide from ethylmagnesium iodide, with subsequent reaction with prop-2-ynyl bromide, a 39.5% yield of 3-prop-2'-ynylindole was obtained. The compound, which was a pale yellow oil slowly becoming darker, was characterised by conversion into 3-indolylacetone
2 by heating it with acidic mercuric sulphate.
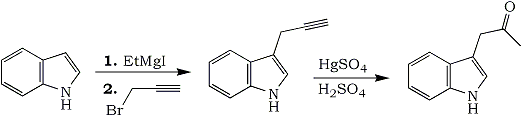 Experimental 3-Prop-2'-ynylindole
Experimental 3-Prop-2'-ynylindoleEthylmagnesium iodide
[from magnesium (4.8 g.) and ethyl iodide (32 g.)] in dry anisole (20 ml.) was cooled in ice, and indole (15.6 g.) in anisole (20 ml.) was added dropwise. After being stirred (0.5 h) at 20°C it was treated at 0°C with prop-2-ynyl bromide (20 ml) in anisole (10 ml) during 15-20 min, stirring continued at 0°C for 11 h, and the mixture then stored at 20°C overnight. It was cooled to 0°C and treated with ether (100 ml), water (200 ml), acetic acid (12 ml), and more water (100 ml), and then extracted with ether (5x25 ml.). The extract was washed with sodium bicarbonate solution and water and dried (MgSO4 and charcoal), the solvents were removed under reduced pressure, and the 3-prop-2'-ynylindole (8.17 g) was distilled as a pale yellow oil, bp 143-145°C/2mmHg, which solidified when cooled below 20°C.
3-Indolylacetone2N Sulphuric acid (10 ml) was treated with mercuric sulphate (0.05 g)
3, stirred and heated on a steam-bath, and the propynylindole (1.5 g) in ethanol (10 ml) added. Stirring and heating was maintained for 2 hr. Pouring the solution into water and treatment with sodium hydrogen carbonate produced a brown gum (1.42 g.), mp 95°C (softening at 75°C). Crystallisation from benzene (charcoal) gave the ketone as brownish rhombs (0.28 g), mp 112-115°C. The mp of a mixture with authentic 3-indolylacetone was 112-115°C.
References[1] Brown, Henbest, and Jones, J. Chem. Soc. 3172 (1952)
[2] Majima and Kotake, Chem. Ber. 55B, 3859-3865 (1922); cf. Kharasch and Reinmuth,
"Grignard Reactions of Nonmetallic Substances", 1954, Prentice-Hall, Inc., New York, p. 81.
[3] Thomas, Campbell, and Hennion, J. Amer. Chem. Soc. 60, 718 (1938)