The chances that tryptophan will cyclisize due to the Pictet-Spengler reaction in the HCHO/NaBH
4 alkylation are IMHO very low, considering the low temperature the reaction is kept at and the small amount of time that is necessary to complete it.
"Pictet-Spengler reactions in aprotic media", Mikolaj Jawdosiuk and James W Cook, J. Org. Chem. (1984), 49, 2699-2701 ,
In 1976 we reported that reaction of tryptophan methyl ester (1) with aldehydes such as benzaldehyde (2a), cyclohexanecarboxaldehyde (2b), or
alpha-keto acids in refluxing benzene (Dean-Stark trap to remove water)
1 provided much improved yields of the Pictet-Spengler reaction with respect to the traditional reaction performed in aqueous, acidic media. The reasons for this were very simple for acid-labile substrates were much less prone to decomposition in a non-acidic, nonaqueous medium.
Since our original reports,
1,2 a number of 3-methoxycarbonyl tetrahydro-
beta-carbolines have been succesfully prepared by this procedure
3,5-7,11. In view of these reports it was suprising to find that Grigg et al. reported that "A repeat of Cook's original work (tryptophan methyl ester, benzaldehyde, benzene, 80°C, 48h), i.e., generating the Schiff's base in situ gave only Schiff's base (1a) and no
beta-carboline (2a,b)"
4.To examine the conflicting experiences regarding this reaction, we have carried out several further experiments.
An important feature of the procedure that was succesful in our hands is use of a Dean-Stark trap below the reflux condensor to remove water from the reaction
1,2.
In the Grigg report, most of the experiments were carried out in sealed NMR reaction tubes, and no mention is made of the use of a water separator.
4 We have compared the course of the reactions of 1 and benzaldehyde (2a, purified by K
2CO
3 wash, drying, distillation) in benzene with an open system and a water separator, and in refluxing benzene in a closed system. Under the former conditions, after 12 h TLC indicated the presence of about 50% imine 4a, the remainder of the material was a mixture of cis and trans carbolines 5a. After 48 h the reaction had proceeded almost completely to 5a. In a closed system without removal of water, the formation of 5a was negligible, and the Schiff's base 4a was recovered quantitatively (Scheme 1).
Scheme 1:
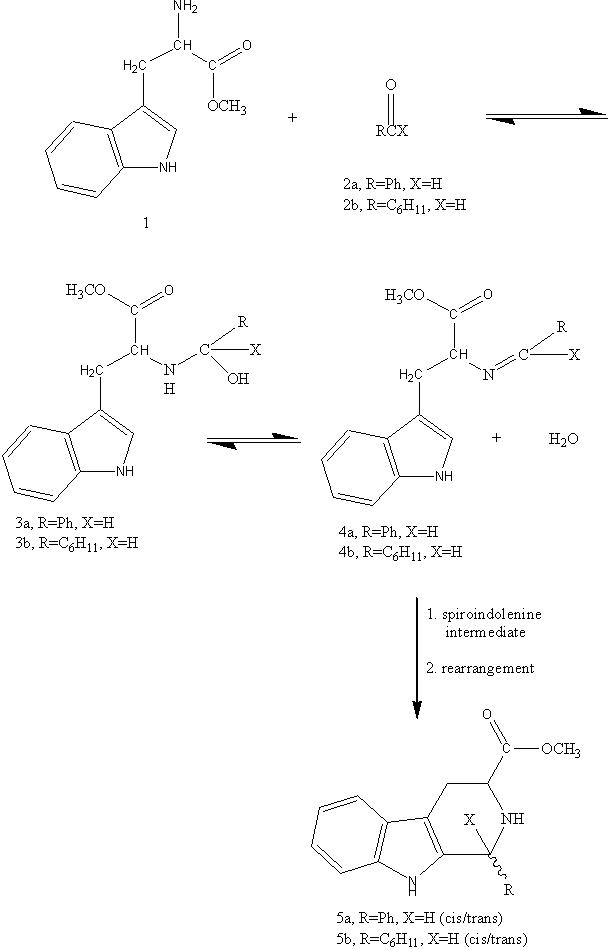
To definitely determine the significance of the use of a Dean-Stark trap in the sequence, identical reactions btween 1 and 2a were performed both open to the air; however, in one case a Dean-Stark trap was used, while in the second experiment none was employed. After 24 h at reflux, aliquots of each reaction were examined by
13C NMR. The condensation performed in the usual manner (DST) gave almost exclusively tetrahydro-
beta-carboline 5a at this point, whereas the reaction carried out in the absence of the trap gave exclusively the non-cyclisized 4a. Nevertheless cyclization in the absence of a DST did eventually take place for after 84 h of heating this gave the cyclized product 5a, but the reaction was extremely slow in comparision to the previously reported conditions (DST).
1,2A second point concerning reaction conditions concerns the presence of acid. Grigg et al. demonstrated the role of acid catalysis in the reaction and suggests that acidic impurities are responsible for succesful cyclization. To test the possibility that air oxidation of aldehyde occurred under the conditions originally used, the reaction of 1 and 2a was carried out in a nitrogen stream, and cyclization to 5a was found to be much slower. Moreover, addition of the proton-scavenger DBU
9 completely suppressed the cyclization. The weaker base imidazole retarded the reaction significantly. Similar results were observed in reactions of 1 with the more reactive cyclohexanecarboxaldehyde (2b). These data support the hypothesis that a small amount of benzoic acid formed by air oxidation facilitates the cyclization.
In regard to the report by Grigg et al.
4 that the ratio of cis and trans isomzers of the 1-phenyl-3-(methoxycarbonyl)-1,2,3,4-tetrahydro-
beta-carbolines (5a) in their series was different from that reported in our earlier work
8, this is entirely correct. The yields in ref 8 (60% trans) were based on isolated material, while examination of the crude product by NMR (see experimental section) indicates the reaction mixture is composed of 5a (58% cis, 42% trans). Hamaguchi reported isolation of a 1:1 of cis to trans isomers of 5a
10. Grigg found this ratio to be 1:33:1 (NMR), while Bhaduri et al.
11 reported that the trans isomer predominated. In the present study a similar ratio (cis > trans) was observed in the 1-cyclohexyl series 5b as well, although on the basis of isolated yields the trans isomer predominated
2.
In conclusion, we emphasise that our experience and that of several other groups
3,5-7,11 establishes, contrary to the implication of Grigg et al,
4 that the simple condensation of tryptophan methyl ester and aldehydes in refluxing benzene in an open system with a water separator provides a simple and very practical method for preparing 3-(methoxycarbonyl)-1,2,3,4-tetrahydro-
beta-carbolines.
Experimental sectionTryptophan methyl ester was obtained from dl-tryptophan by esterification in methanolic HCl and converted into the freebase (K
2CO
3).
12 This material 1 was recrystallised from ether-hexane (mp. 70.5-72°C (lit. mp.
13 71-73°C)). NMR spectra were recorded on Varian T-60 MHz, Varian CFT-20
13C NMR and Bruckner WH-250 (250 MHz Fourier transform instrument with multinuclear capability) spectrometers. Precoated TLC sheets used were silica gel 60F-254 (E. Merck). TLC plates were developed with the spray reagent ceric ammonium sulfate in 50% sulfuric acid.
Reactions of tryptophan methyl ester (1) with benzaldehyde (2a). Preparation of Nb-Benzylidenetryptophan methyl ester (4a).Tryptophan methyl ester (1, 4.4 gr, 0.02 mol) and benzaldehyde (2a, 2.5 gr, 0.025 mol) were dissolved in benzene (70 mL) and the solution was refluxed for 1 h in a flask equipped with a Dean-Stark trap and a reflux condensor open to air. The solvent was evaporated and the residue was recrystallised from metrhanol to afford 5.87 gr material 4a which melted at 123-126°C (95.7% yield). An analytical sample melted at 130°C (lit. mp. 120°C
1, 128-129°C
4);
13C NMR (CDCl
3, Me
4Si) 29.7, 52.1, 73.8, 111.2, 118.8, 119.4, 122.0, 123.6, 127.4, 128.5, 131.0, 135.8, 136.3, 163.3, 172.7.
Direct preparation of 3-(methoxycarbonyl)-1-phenyl-1,2,3,4-tetrahydro-9H-pyrido[3,4-b]indole (5a) from 1 and 2a1. Tryptophan methyl ester (1, 2.2 gr, 0.01 mol) and benzaldehyde (2a, 1.1 gr, 0.0105 mol) from a newly purchased bottle were dissolved in distilled benzene (50 mL). The solution was held at reflux under a reflux condensor (open to air) equipped with a Dean-Stark trap. The reaction progress was monitored by TLC after 12, 24, 48 and 70 h by comparision of Rf values with those of authentic samples
2. After 12 h about 50% of the Schiff base was converted into a cis/trans mixture of 5a. After 24 h the conversion increased to about 70-85% (estimated by TLC). After 48 hours the reaction mixture showed only a small amount of unreacted 4a (TLC) and after 70 h the reaction was essentially completed. Evaporation of benzene to dryness gave 3.19 g of crude 5a. TLC of this material showed only two spots corresponding to the cis and trans isomers of 5a. The chemical shifts of the carbon atoms in the
13C NMR spectrum of the material were consistent with those reported in the literature
8. The crude product was dissolved in CDCl
3 and the
1H and
13C NMR spectra were taken. The integration of the C(1) protons in the cis and trans 5a isomers (250 MHz NMR spectrometer) was consistent with a ratio of cis to trans of 58:42 while integration of the
13C NMR spectrum with suppressed NOE for several superimposable resonance lines also gave the value of 58:42. These values are consistent with values reported in the literature (54:46) measured with mass spectroscopy
3 and (54.5:45.5)
4 measured by 90 MHz
1H NMR.
2. The reaction described above (see 1) was carried out in an open system exactly analogous to the above experiment but in absence of a Dean-Stark trap (water separator). After 24 h an aliquot of the reaction mixture was analysed by
13C NMR and was shown to essentially contain only the imine 4a. On heating the mixture for 36 h the tetrahydro-
beta-carbolines began to appear (TLC +- 30% 5a) and after 84 h 4a had cyclized completely to 5a. The control experiment (experiment 1, DST) after 24 h at reflux contained essentially only 5a (
13C NMR analysis).
3. The same quantities of 1 and 2a and benzene were used as in the two previous cases, however, 2a was washed with a 50% solution of K
2CO
3 and then dried (MgSO
4) and distilled under reduced pressure at 38°C (1 torr). The reaction mixture was held at reflux for 84 h in a closed system with a reflux condensor in the absence of a Dean-Stark trap. The condensor was closed with an oil bubbler. After 84 h TLC did not show any appreciable amounts of the cyclized product 5a. Evaporation of benzene to dryness gave a qsuantitative recovery of 4a.
4. The reaction sequence termed 3 above was repeated by employing a Dean-Stark trap but under a nitrogen (Ameri-Gas 99%) atmosphere. After the reaction was held for 12 h in refluxing benzene, no trace of 5a was observed (compare 50% in experiment 1), and after 48 h only 10% of 5a had been formed.
5. The reaction sequence termed 1 as above was carried out in the presence of 1.5 g (0.01 mol) of DBU (1,8-dizazbicyclo-[5.4.0]undec-5-ene) although 2a was pretreated as indicated under 3. After 70 h no 5a was detected. Although DBU is a strong enough base
9 to act as a proton scavenger and also abstract proton from the
alpha position of the ester, the Schiff base was observed in this sequence by TLC.
The same reaction was repeated (reagents the same as above) with addition of 0.7 g (0.01 mol) of imidazole. After the mixture was held for 50 h in refluxing benzene, the amount of cyclized product 5a was estimated to be 10-15%
References1: Sandrin J et al,
Heterocycles 4 (1976), 11012: Soerens, D et al,
J. Org. Chem. (1979) 44, 5353: Kumar, S; Seth, M; Bhaduri A P,
Ind. J. Chem. B (1981) 10784: Grigg R et al,
J. Chem. Soc. Perkin Trans. I (1983) 1855: Harisson, D M ,
Tetrahedron Lett. 22, (1981) 25016: Shimizu, M et al,
Chem. Pharm. Bull. 30 (1982) 34357: Toyoda, Y et al,
Chem. Lett. (1982) 9038: Ungemach, F et al,
J. Am. Chem. Soc. 102 (1980) 69769: DBU has been employed to deprotonate malonic esters for alkylation reactions and therefore should serve as a good proton scavenger in this case (Oedigerod, H and Moeller, F,
Liebigs Ann. Chim. (1976) 348). It is also a strong enough base to deprotonate trinitrotoluene (Sugimoto, N. et al,
J. Phys. Chem. 86 (1982) 3418; Ebel, H, F. C-H Acidity of organic compounds in "
Methods der Organischen Chemie, Houben-Weyl, 1970, 13 (part I), 27, 57).
10: Hamaguchi, F; Nagasaka, T; Ohki, S,
Yakugaku Zasshi (1974) 94, 351 11: Kumar, S et al,
Ind. J. Chem. (1983) 22B, 5412: Otsuka, H & Inouye, K,
Bull. Chem. Soc. Jpn. 37 (1964) 146513: Brenner, M; Sailer, E; Kocher, V,
Helv. Chim. Acta 31 (1948) 1908