Ups, the hydrogen flas was not turned off, that's why no presuredrop was seen. Here is the writup
(To Rhodium, iff you wan't a HTML I mail you one)
From aldehyde to alcohol
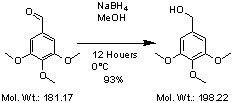
50g(0,28mol) 3,4,5-trimethoxybensaldehyde was dissolved into 600mL 0°C methanol (ice bad) under stirring. Then 10g NaBH4 was added in 3 portions, the reaction was left over night.
The next day most of the methanol was removed (rotorvap, important to get a nice extraction)) and the rest diluted with water (500mL) and made acid with 37% HCl(aq). The water was extracted with 4*100mL CH2Cl2, the organic phases was combined, dried and concentrated on the rotor vapor.
Yield 47,2g(93%) 3,4,5-trimethoxybenzylakohol.
Rf (40% EtOAc in Heptane) 0,27
From alcohol to bromide

47,2g(0,24mol) 3,4,5-trimethoxybenzaldehyde was dissolved into 1L ether at 0°C (ice bad) under stirring. Then 25g(0,1mol) PBr3 was added over 30min, the ice removed and the reaction left for 60 min. After that the mixture was poured out into 300mL water, the organic phase was separated, the water extracted with 2*100mL ether, the ether phases combined, washed with 2*50mL water, dried and concentrated (rot. vap.)
Yield 55g(88%) 3,4,5-trimethoxybenzylbromice
Rf (50% EtOAc in Heptane) 0,63
From bromide to nitile
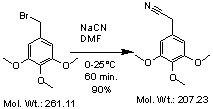
50g(0,19mol) 3,4,5-trimethoxybenzylbromide was dissolved into 600mL DMF under stirring at 0°C (ice bad), then 20g NaCN vas added and the ice bad removed. After 1 hour the mixture was poured out into 2L water, the water was extracted with 4*100mL CH2Cl2. The organic phases was combined, washed with 2*100mL water, dried and concentrated.
Yield of 3,4,5-trimethoxybenzylcyanide 36g(90%)
Rf (50% EtOAc in heptane) 0,45
From nitrile to ammine

15g(0,07mole)3,4,5-trimethoxybenzylcyanide was dissolved in 100mL ethanol, 10mL 37% hydrochloric acid and 100mg 10% Pd/C was added and the mixture was hydrogenated at 5,5bar for 12 hours, then the mixture was filtrated, most of the ethanol was evaporated off, then the mixture was dissolved into 200mL water, extracted with 50mL ether (to remove starting material ect.), made basic with NaOH, extracted with 3*40mL CH2Cl2, the organic phases was combined, dried and evaporated.
Yield 13,7g mescaline freebase.