Now you ask: What does anybee want with 1,4-dimethoxy-2-nitrobenzene? The nitro group itself is pretty useless but reduction to the 2,5-dimethoxyaniline can bee done with lots of reagent systems (Fe/HCl to mention the most easy one) and this opens lots of synthetic possibilites via diazonium intermediates. E.g.
2C-T-X precursors via
Post 473295 (missing)
(azole: "ArNH2 --> ArSMe via aryldiazonium + CuSMe", Novel Discourse)2,5-dimethoxy-P2P via
Post 448826 (missing)
(Lego: "P2Ps via Meerwein arylation Actually Lego was...", Methods Discourse)2,5-dimethoxybenzaldehyde via
Post 355321
(foxy2: "formylation by the "nitroso" method", Novel Discourse)2-Cl/Br/I/CN-1,4-dimethoxybenzene via Sandmeyer reaction
Of course 2,5-dimethoxy-4-nitrotoluene can bee used as a DOM precursor.....
For our ADHS-bees see the practical, short version below

J. Chem. Res. (S), 2000, 106-107The dichotomy between nitration of substituted 1,4-dimethoxybenzenes and formation of corresponding 1,4-benzoquinones by using nitric and sulfuric acidC. Waterlot, B. Haskiak and D. Couturier
Université des Sciences et Technologies de Lille, Laboratoire d’Ingéniérie Moléculaire,
Bâtiment C4, ler étage 59655 Villeneuve d’Ascq Cedex, France
Abstract: Various alkyl-substituted p-dimethoxybenzenes (ArH) react readily with nitric acid and sulfuric to form nitroproducts (ArNO
2). When the nitric acid is used in excess, the nitro-product react via either nitration to dinitrocompound (Ar(NO
2)
2) or via oxidative demethylation to nitro- p-quinone (Q). As such, the competition between the nitration, polynitration and oxidative dealkylation is effectively modulated by the added nitric acid and the alkyl-substituted p-dimethoxybenzenes.
Aromatic nitration is conventionally carried out with nitric acid, either alone or in combination with either Lewis or Brönsted acid
1. It has long been known that most of the aromatic compounds are nitrated with a mixture of nitric acid and sulfuric acid according to ionic mechanism.
2 For compounds as (polymethoxy)benzenes, more reactive than toluene, it has been shown that nitric acid was a sufficient nitration reagent to nitrate them.
1 Only in this case, it has been suggested that the nitration of the (polymethoxy)benzenes with nitric acid occurs via a radical pathway (Scheme 1.)
3,4 In the meantime, other nitration reagents have been studied for nitration of activated-arenes.
5 It has been proved that the mechanism given for the nitration reactions with NO
2 and HNO3 (Scheme 1) is generally valid for the nitration of donoractivated benzenes with nitric acid.
6 Nevertheless, the mechanism is not clear in all details and it is still under discussion.
1,4-8In connection to our interest in deoxygenation of dissolved oxygen in water, we investigated the nitration of dimethoxybenzene derivatives with a mixture of nitric and sulfuric acid. For the first time, nitration and oxidative demethylation of (polymethyl)-1,4-dimethoxybenzenes were observed in the studied conditions. On the other hand, because the tetramethyl-p-dimethoxybenzene is oxidatively demethylated by nitrogen dioxide to form duroquinone,
9,10 we thought that it could be interesting to examine the dichotomy between aromatic nitration and quinone formation by using (polymethyl)-1,4-dimethoxybenzenes
1 (Scheme 2) with an excess of nitric acid (molar ratios : HNO
3/H
2SO
4 = 1.5/1.1).
Results and discussionOur starting point was to consider that the nitration of 1,4-dimethoxybenzene derivatives with nitric acid followed the radical reaction scheme to form quinone-type compounds by oxidative demethylation if reactions occurred in weakly acidic medium. We then investigated the nitration of (mono or polymethyl)-1,4-dimethoxybenzenes with a mixture of nitric and sulfuric acid in acetic acid (Scheme 2). Several experiments were performed at various temperatures and times, in order to find the best conditions to synthesize nitro-compounds
2 without any side products. The results are summarized in Table 1.
Results show that the nitration of aromatic compounds
1 is more important than the oxidative demethylation (Table 1, entries 2, 4, 6, 10 and 12). On the other hand, the dichotomy is less pronounced in the studied conditions (polar solvent) than Kocki’s system
10 (entry

. By using H
2SO
4, the first corresponding step is the formation of NO
2+ to give a cation intermediate (s-complex) and then, the electrophilic substitution of the donor-arenes to form the compounds
2 (Scheme 3, path A).
9 However, we cannot exclude by the presence of traces of nitrous acid in nitric acid, the formation of NO
2 and so the reaction between NO
2· and so the reaction between NO2· with the radical cation
1+ to give the s-complex (Scheme 3, path B).
4,8In a second step, the chemical behaviour of nitro-(mono or polymethyl)-1,4-dimethoxybenzenes
2 towards nitric acid in excess is different. Thus, the reaction of 2-nitro-1,4-dimethoxybenzene (Table 1, entry 2) with HNO
3/H
2SO
4 produce the 2,5-dinitro-1,4-dimethoxybenzene. On the other hand, tetramethyl-1,4-dimethoxybenzene (Table 1, entries 13 and 14) react with HNO
3/H
2SO
4 to produce p-benzoquinones via oxidative demethylation.
10 Other products are obtained by nitration and oxidative demethylation of certain p-dimethoxybenzene derivatives (Table 1, entries 2, 8 and 12). According to previous work,
4,6,8 the reaction of compounds
2 with an excess
of HNO
3 to form nitro-benzoquinones derivatives proceeds probably via a radical pathway but we cannot confirm if the radical
2· is generated by electron transfer between the arenes
2 and NO
2+4 or by electron transfer from the arenes
2 to NO
+8.
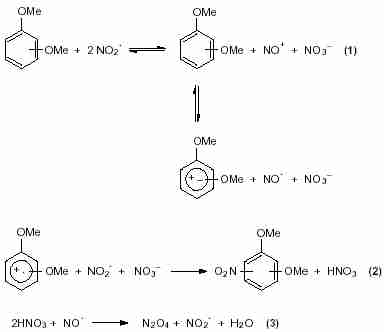 Scheme 1
Scheme 1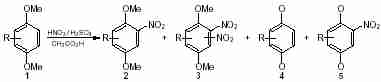 Scheme 2
Scheme 2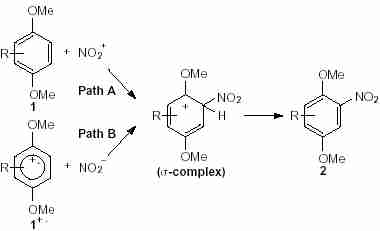 Scheme 3
Scheme 3