Synthesis and Pharmacological Characterization of a Series of Geometrically Constrained 5-HT2A/2C Receptor LigandsJames J. Chambers, Jason C. Parrish, Niels H. Jensen, Deborah M. Kurrasch-Orbaugh, Danuta Marona-Lewicka, and David E. NicholsJ. Med. Chem. 46 (16), 3526 -3535 (2003)
(
https://www.thevespiary.org/rhodium/Rhodium/pdf/nichols/nichols-naphtofurans-2.pdf)
DOI:
10.1021/jm030064v S0022-2623(03)00064-5
 Abstract
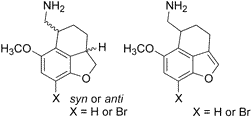
AbstractIn studies of the SAR of phenethylamine-type serotonin 5-HT2A receptor agonists, substituted conformationally constrained tetrahydronaphthofurans were designed to investigate the optimal conformation of the 2-aminoethyl moiety. These compounds were tested using in vitro assays for affinity at 5-HT1A, 5-HT2A, and 5-HT2C receptors. The benzofuran-containing analogues,
6a and
6b, had significantly higher affinity for the 5-HT receptors tested than did the benzodihydrofuran-containing compounds,
4a,
4b,
5a, and
5b. The most potent compound (8-bromo-6-methoxy-4,5-dihydro-3H-naphtho [1,8-bc]furan-5-yl) aminomethane,
6b, had
Ki values for displacement of [
125I]-DOI from 5-HT2A and 5-HT2C cloned rat receptors of 2.6 and 1.1 nM, respectively. Despite their high affinity, the compounds of this naphthofuran series lacked high intrinsic activity at the 5-HT2A receptor as measured using the phosphoinositide hydrolysis assay. The most potent compound in vitro,
6b, was tested in the two-lever drug discrimination assay in rats trained to discriminate LSD from saline, and failed to substitute, a result typical for compounds with low intrinsic activity. Thus, although conformational constraint has led to high-affinity 5-HT2A ligands with partial agonist activity, all of the spatial and steric properties of the ligand necessary for full receptor activation have not yet been identified.