would the absesnce of literature references then indicate that wolff-kishner style reduction would not work in this case?Yes, unfortunately, that is most often the case, at least when it comes to simple molecules like these.
I have however found some one-step deoxygenations of cinnamaldehyde to propenylbenzene/allylbenzene using cyanoborohydride with a lewis acid additive, which also seems to deoxygenate benzaldehydes to methylbenzenes and aralkyl ketones to hydrocarbons in high yield:
Chemoselective reductive deoxygenation of  ,
,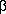 -unsaturated ketones and allyl alcoholsA. Srikrishna, R. Viswajanani, J. A. Sattigeri and C. V. Yelamaggad
-unsaturated ketones and allyl alcoholsA. Srikrishna, R. Viswajanani, J. A. Sattigeri and C. V. YelamaggadTetrahedron Letters 36(13), 2347-2350 (1995)
(
https://www.thevespiary.org/rhodium/Rhodium/pdf/cinnamaldehyde2propenylbenzene.pdf)
DOI:
10.1016/0040-4039(95)00252-8
AbstractA simple and convenient procedure for a highly chemoselective reductive deoxygenation of

,
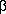
-unsaturated ketones and allyl alcohols to olefins by sodium cyanoborohydride and boron trifluoride etherate in dry THF is described.
 Reductive, selective deoxygenation of acylbenzofurans, aromatic aldehydes and ketones with NaBH3CN-TMSClVernon G. S. Box and Panayiotis C. Meleties
Reductive, selective deoxygenation of acylbenzofurans, aromatic aldehydes and ketones with NaBH3CN-TMSClVernon G. S. Box and Panayiotis C. MeletiesTetrahedron Letters 39(39), 7059-7062 (1998)
(
https://www.thevespiary.org/rhodium/Rhodium/pdf/deoxygenation.nabh3cn-tmscl.pdf)
DOI:
10.1016/S0040-4039(98)01519-6
AbstractAromatic aldehydes, ketones and acylbenzo[b
]furans have been reductively deoxygenated with sodium cyanoborohydride and the mild electrophile chlorotrimethylsilane.

A Simple and Convenient One Step Method for the Reductive Deoxygenation of Aryl Ketones to Hydrocarbons
Srikrishna, A.; Sattigeri, J. A.; Viswajanani, R.; Yelamaggad, C. V.
Synlett, (1) 93-94 (1995)
(https://www.thevespiary.org/rhodium/Rhodium/pdf/deoxygenation.nabh3cn-bf3-et2o.pdf)