A Concise and Stereoselective Synthesis of (±)-erythro-MethylphenidateDennis Russowsky, and Brenno Amaro da Silveira NetoTetrahedron Letters, 44(14), 2923-2926 (2003)
(
https://www.thevespiary.org/rhodium/Rhodium/pdf/erythro-methylphenidate.pdf)
DOI:
10.1016/S0040-4039(03)00420-9
 Abstract
AbstractA concise and stereoselective synthesis of racemic erythro-methylphenidate (
1) is described. The coupling reaction between piperidine-2-thione (
3) and 2-bromo-2- phenylmethylacetate (
4) afforded the
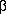
-enaminocarbonyl compound
2 in 60% yield by a modified Eschenmoser sulfide contraction reaction. In most cases the bicyclic thiazolidinone
5 was produced. Diastereoselective reduction of
2 in the presence of borohydrydes furnished the (±)-erythro-methylphenidate in good yields with
dr >95%.