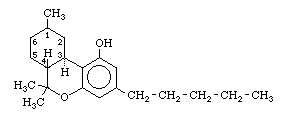
Under the term tetrahydrocannabinol (THC) there are three known physiologically active isomers, which differ only in the bonding in the left-hand ring of the molecule:
The preferred numbering for the isomers is based on the mono-terpene numbering scheme
[1] (used here) although other systems are used and confuse the literature
[2]. The isomers have been designated here as follows;
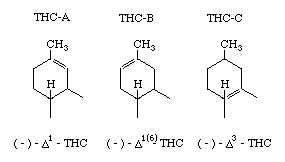
THC-A's can be easily isomerized-to THC-B's by heating with p-toluene sulphonic acid in nearly quantitative yield
[3]. The syntheses of THC-A, THC-B, or THC-C are all based on the condensation of olivetol (5-(n-amyl)-resorcinol) with a second component which forms the left-hand and center ring of the THC molecule. THC-A or THC-B are obtained from olivetol and one of the following; citral, (-)-verbenol, or (+)-p-2,8-menthadien-2-ol. THC-C is obtained from olivetol and (-)-2-carbethoxy-5-methylcyclohexane.
References:[1] http://www.acdlabs.com/iupac/nomenclature/79/r79_88.htm
[2] http://www.mikuriya.com/s5_6.pdf
[3] JACS 90, 2420 (1968)