From Walton's
Inorganic Laboratory PreparationsBoron TrifuorideBoron trifluoride, BF3, is a gas remarkable for the many addition compounds it forms with compounds of oxygen, nitrogen, sulfur, and other elements-even with the inert gases. The electronic structure shows that the electron shell of boron is incomplete, with room for another pair of electrons. That is, it is an acceptor molecule, and its addition compounds are all formed by donation of a pair of electrons from the other molecule to form a coordinate link with the boron. They are among the simplest types of coordination compound. Because of its electron-accepting power, boron trifluoride is an excellent catalyst for many reactions such as alkylations and polymerizations. It is prepared in considerable quantities for use in the petroleum and synthetic organic chemical industries.
Boron trifluoride is similar in many ways to silicon tetrafluoride, including its physical properties and reactions with water and alkalies. It could be prepared by an analogous method. However, a smoother reaction which gives a purer product, less contaminated by silicon tetrafluoride, this reaction will be used here. First, boric oxide and ammonium fluoborate must be prepared, since these are not generally available. (Any soluble fluoborate could be used in place of the ammonium salt.)
Boric oxide Heat 30 grams of boric acid in an iron dish over a Meker burner, stirring occasionally with an old file, until a clear viscous melt is obtained. Cool quickly, break out the glassy mass, crush it in a rock crusher or other device (it is very hard and brittle, so do not attempt this preliminary crushing in an ordinary mortar ; then grind to a powder in a large iron mortar.
Ammonium fluoborate Grind up 65 grams (1.15 moles) of ammonium acid fluoride, NH4HF2, with 30 grams (0.5 mole) of boric acid, and heat the mixture in a shallow iron dish over a gas ring or large burner flame in the hood. Frothing occurs, ammonia and steam escape, and the excess of NH4HF2 is volatilized. Continue to heat until a dry, puffed-up white mass remains. Break this up with a pestle and heat again, but stop as soon as dense white fumes indicate that the main product is starting to sublime. Cool; then powder in a mortar. This product is pure enough for use in preparing boron trifluoride. If a clean, pure preparation of ammonium fluoborate is required for another purpose, the best way to make it is to mix the theoretical amounts of ammonium acid fluoride and boric acid in aqueous solution in a plastic dish, and evaporate to crystallization on a steam bath.
 Boron trifuoride
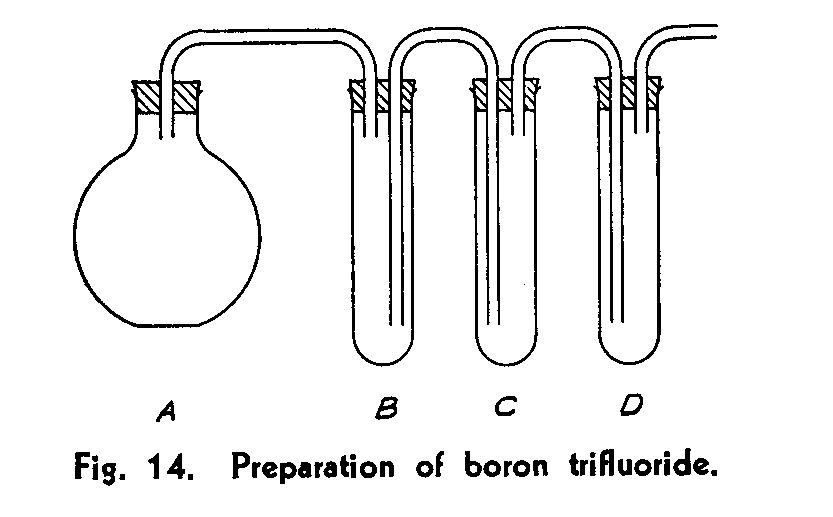
Boron trifuoride Set up in the hood the apparatus shown in Fig. 14. Since boron trifluoride attacks cork less than it does rubber, cork stoppers should be used and rubber connections avoided as far as possible. (Where a very pure product is desired, an all-glass apparatus must be used. Ground joints may be lubricated with a paraffin-vaseline mixture.) The vessels and connections should be of Pyrex glass.
The generating flask A has a volume of 500 ml. The test tubes B, C, and D are 25 X 200 mm. B is empty and used as a trap; C contains some 30 ml of sulfuric acid saturated with boric acid, to absorb hydrogen fluoride from the gas stream. In A, place the entire product of ammonium fluoborate, well mixed with 6 grams of finely powdered boric oxide. In D, place 30 ml of diethyl ether, and cool the tube in ice. Arrange for the gas passing out of D to be sucked away by the draft or to be absorbed in caustic soda.
When all is ready, pour 40 ml of concentrated sulfuric acid into flask A and replace the cork immediately. A vigorous reaction occurs, which, however, soon slackens and must be maintained by heating. Keep the reaction going steadily but not too fast. Some of the boron trifluoride is absorbed by the ether in D, forming a viscous solution of the addition compound (C2H5)20•BF3. This will be used for the tests to follow.
Before all the boron trifluoride has been generated, replace the test tube of ether with a test tube containing a few milliliters of water. Note the reaction between boron trifluoride and water. If sufficient gas is passed, viscous liquids BF3-21120 and BF,-H20 are obtained. These liquids are strongly acidic, the latter ionizing to H+ and HO•BF3 . Similar compounds are formed from boron trifluoride and alcohols.
Pass a little boron trifluoride gas into a Bunsen flame. Note the characteristic color, which is given by all volatile boron compounds and is a sensitive test for boron.
TestsPerform the following tests on the boron trifluoride-ether solution:
1. Pass ammonia gas into 10 ml of the solution. The ammonia can be drawn from a cylinder or prepared by dropping concentrated aqueous ammonia onto sodium hydroxide pellets and drying the gas by contact with potassium hydroxide sticks or pellets. The white precipitate is the complex NH3•BF3. Filter the solid on a Buchner funnel, wash it with a little ether, and allow it to dry. Heat the solid in a dry test tube; it sublimes unchanged.
2. Mix a few drops of the solution with a few drops of pyridine. Note that a similar addition product is formed.
3. In a small distilling flask mix 10 ml of the solution with 12 ml of acetic anhydride. Reflux the mixture for 10 minutes, then distill slowly, collecting the distillate which comes over between 65 and 100°. It should be mainly ethyl acetate.
Wash the distillate with about 50 ml of 10 per cent sodium carbonate to free it from boron trifluoride and any acetic anhyride; collect the ethyl acetate with a separatory funnel, noting its smell and its volume. The yield would be practically theoretical, except that some of the ethyl acetate remains behind in the distilling flask as the very stable complex CH3C0002H6•BF3.
Make the following test with the aqueous solution of boron trifluoride: Test for fluoride ion by diluting with water, neutralizing with sodium bicarbonate, making slightly acid with acetic acid, and adding calcium chloride solution.
A striking test of the catalytic power of boron trifluoride can be made if the materials are available. Condense some isobutylene in a beaker surrounded by dry ice, or simply put some pieces of dry ice in a beaker and pass in gaseous isobutylene; then add a few drops of isoprene. Stir the liquid mush and pass in boron trifluoride. Immediately a solid white mass of butyl rubber is formed.9
7 Inorganic Syntheses, 1, 21, and 2, 23.
F. J. Sowa and J. A. Nieuwland, J. Am. Chem. Soc., 58 (1936), 272.
8 Inorganic Syntheses, 2, 23.
9 Annual Reports of the Chemical Society, 1942; p 128
