I think you guys are barking up the wrong tree. I do not see how the USP monograph is going to help you. However I will post the monographs if you like. Pseudoephedrine has quite a few monographs. Of the monographs in the pseudoephedrine folder which ones would you like to see?
Pseudoephedrine HCL
Pseudoephedrine Extended Release Capsules
Pseudoephedrine Hydrochloride Oral Solution
Pseudoephedrine HCL Syrup
Pseudoephedrine HCL Tablets
Pseudoephedrine HCL Extended Release Tablets
Pseudoephedrine Sulfate
Dissolution Apparatus:
I am unlcear on how this information will be of value to you either however someone asked for it.
Apparatus 1— The assembly consists of the following: a covered vessel made of glass or other inert, transparent material1 ; a motor; a metallic drive shaft; and a cylindrical basket. The vessel is partially immersed in a suitable water bath of any convenient size or placed in a heating jacket. The water bath or heating jacket permits holding the temperature inside the vessel at 37 ± 0.5 during the test and keeping the bath fluid in constant, smooth motion. No part of the assembly, including the environment in which the assembly is placed, contributes significant motion, agitation, or vibration beyond that due to the smoothly rotating stirring element. Apparatus that permits observation of the specimen and stirring element during the test is preferable. The vessel is cylindrical, with a hemispherical bottom and with one of the following dimensions and capacities: for a nominal capacity of 1 liter, the height is 160 mm to 210 mm and its inside diameter is 98 mm to 106 mm; for a nominal capacity of 2 liters, the height is 280 mm to 300 mm and its inside diameter is 98 mm to 106 mm; and for a nominal capacity of 4 liters, the height is 280 mm to 300 mm and its inside diameter is 145 mm to 155 mm. Its sides are flanged at the top. A fitted cover may be used to retard evaporation2. The shaft is positioned so that its axis is not more than 2 mm at any point from the vertical axis of the vessel and rotates smoothly and without significant wobble. A speed-regulating device is used that allows the shaft rotation speed to be selected and maintained at the rate specified in the individual monograph, within ±4%.
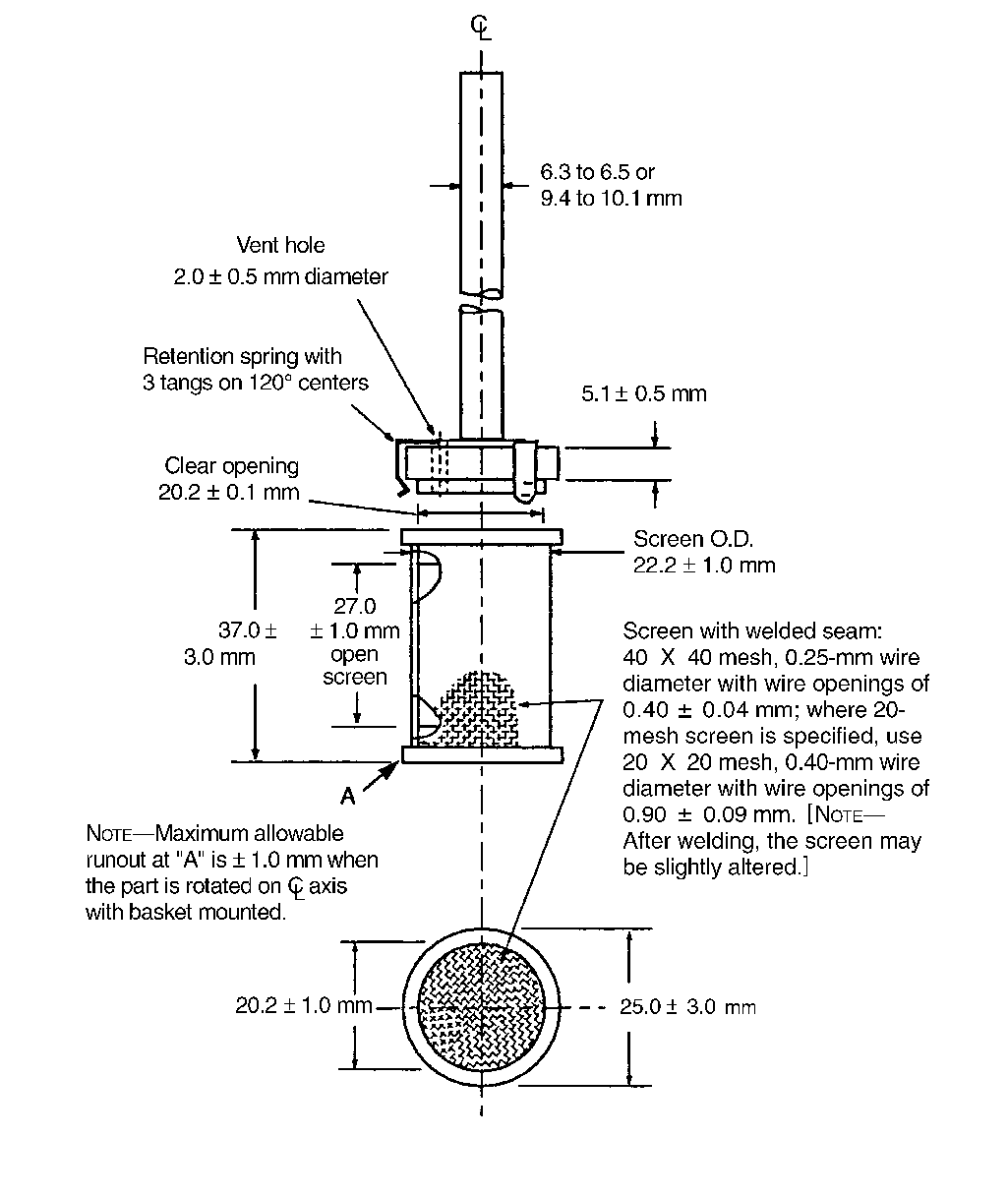
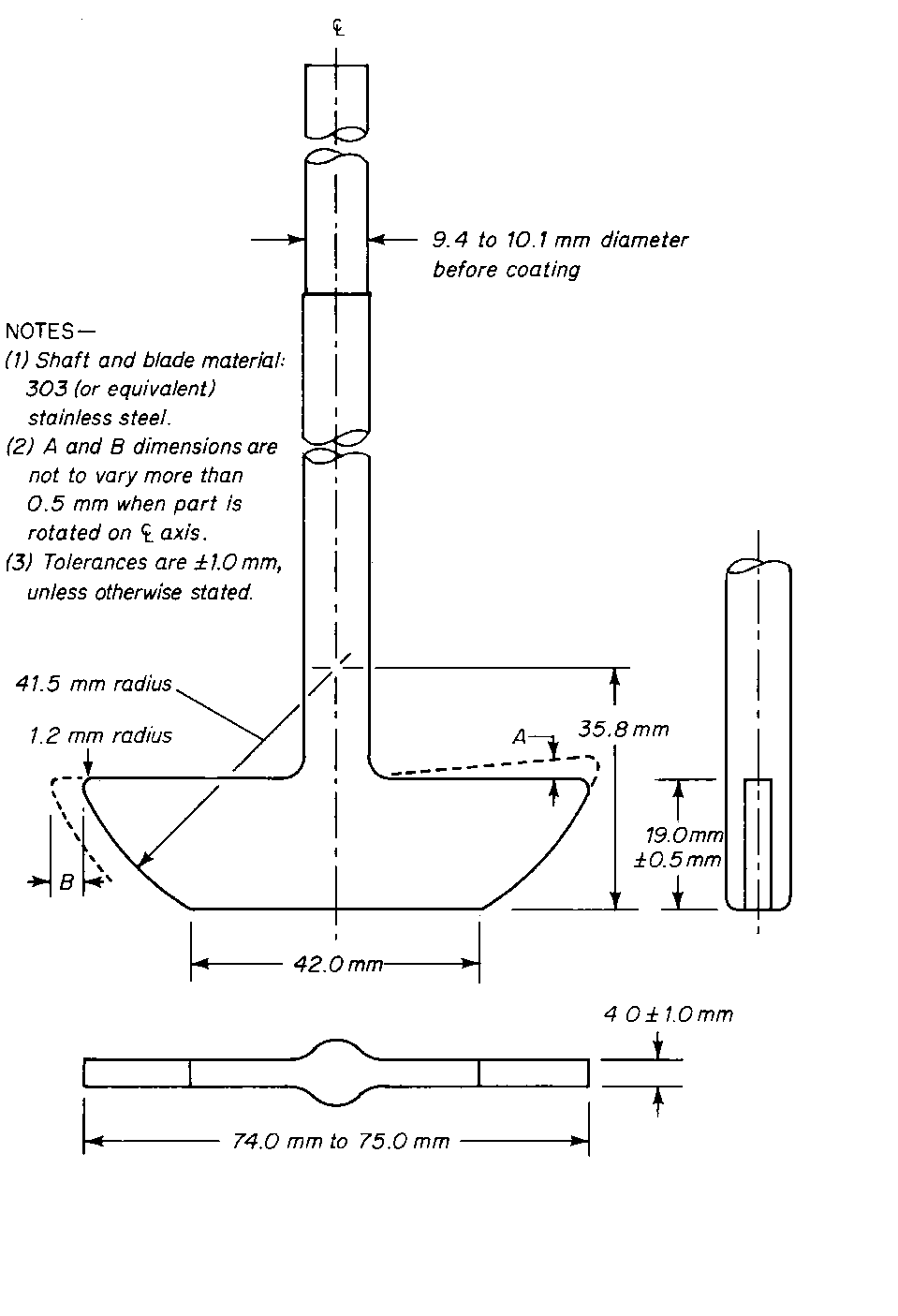
Apparatus 2— Use the assembly from Apparatus 1, except that a paddle formed from a blade and a shaft is used as the stirring element. The shaft is positioned so that its axis is not more than 2 mm at any point from the vertical axis of the vessel and rotates smoothly without significant wobble. The vertical center line of the blade passes through the axis of the shaft so that the bottom of the blade is flush with the bottom of the shaft.
regards,
m_e