Post by: n00dle on August 25, 2004, 02:49:00 PM
Getting woodrose seeds,grinding, defatting them, extracting with ethanol and purifying either by recrystallization or chromatography to pure LSA, dumping them in some KOH soln to hydrolyse to lysergic acid, making some diethylamine from DEET + NaOH and coupling the diethylamine and lysergic acid with some BOP reagent (or PyBOP since this is non-toxic) or HATU or some other coupling reagent to yeild LSD. Since the only reaction is the BOP reaction and these are touted to yeild high in room temperature in a couple of hours, the yeild should be immensely higher than the old-style multiple-rxn synthesis's.
Why does this seem -too- easy? What's the catch? Everything is OTC, and PyBOP doesnt have any suspicion around it.
It's too easy..
Post by: armageddon on August 25, 2004, 05:33:00 PM
extracting with ethanol and purifying either by recrystallization or chromatography to pure LSA, dumping them in some KOH soln to hydrolyse to lysergic acid, making some diethylamine from DEET + NaOH
purifying LSA from plant matter is a bitch to work with unless you use column (or other) chromatography; KOH hydrolysis has to be done carefully (as you know, LSA isn't that stable), then there's the isomerisation/rotation question, and I don't know about making diethylamine from DEET (what's that?), maybe it is easy to perform, maybe not.
The coupling reaction sounds good though, but then again: where to obtain the coupling reagent from? Synthing your own isn't that easy either (peptide chemistry is a chapter in itself).
My personal favorite is the reaction described here (
Post 412547 (https://www.thevespiary.org/talk/index.php?topic=12738.msg41254700#msg41254700)
(pHarmacist: "Synthesis of LSD from Lysergic Acid", Tryptamine Chemistry), as it starts with (optically pure) lysergic acid too (pretty simple: defat/extract woodrose seeds, chromatograph and isolate optically active isomers via tartrate), and promises acceptable yields with using easy chemicals (DMF, diethylamine and CDI and obviously that gas cylinder of argon/helium/N2 or whatever) and affordable equipment. Only thing that could bee difficult is getting hold of some carbonyldiimidazole. Doesn't sound very common!? (any ideas for synthing your own?)Seems like the problem with those -too- easy looking LSD synths is obtaining the needed catalysts/coupling reagents IMO.
Greetz A
Post by: SpicyBrown on August 25, 2004, 11:16:00 PM
It seems to me that with lysergic acid however, intermolecular reactions between one's carboxylic acid and the other's indole amine might be a problem. How does the reactivity of the indole amine compare to the reactivity of diethylamine?
Anyway, I think something like EDC might work better then an activating agent like HBTU, at least in ease of post-reaction purification. The first wall to get by though really is if one could hydrolyze the LSA's amide easily without blowing it all to hell.
-SpicyBrown
Post by: n00dle on August 26, 2004, 08:23:00 AM
purifying LSA from plant matter is a bitch to work with unless you use column (or other) chromatography
Of course, but we're talking two chromatographs per the entire synthesis for a product which makes it worth it. If someone was too lazy to not purify thier product/precursors then they shouldn't be messing around with this kind of stuff.
KOH hydrolysis has to be done carefully (as you know, LSA isn't that stable)
LSA hydrolysis information was taken off Rhodium's page.
"Lysergic Acid From Ergot Alkaloids.
Dissolve 20 g of the alkaloid (use any of the above or one of its isomers or a combination) in 200 ml of 1 M methanolic KOH solution (this is made by dissolving 14 g of KOH in 250 ml of dry methanol) in a 1 1iter evaporation flask (heavy walled construction). Evaporate the methanol off. Add 400 ml of 8% aqueous (water) KOH solution to the residue and boil for one hour under a slow stream of nitrogen that is allowed to flow through a small orifice for exhausting purposes. Cool, acidify with dilute sulfuric acid, and shake in a separatory funnel with 1 1iter of dry ether. Separate the lower aqueous layer and filter it with vacuum assist. Wash the precipitate with 20 ml of dilute sulfuric acid. This is lysergic acid; store as described later in this chapter."
It sounds like someone's already done this..
then there's the isomerisation/rotation question
Seperation: Precipitate the iso-lysergic acid by adding some 10% HNO3, filter, add more portions until no more precipitate forms. Convert it to lysergic acid by adding 3 ml of 10% KOH per every 0.1 g of iso-lysergic acid, heat on steam bath for 1 hour under a nitrogen atmosphere. Precipitate the changed lysergic acid by acidifying with glacial acetic acid.
The only annoying thing in that is the steam bath/nitrogen atmosphere, but still, it gets your iso-lysergic acid to lysergic acid.
I don't know about making diethylamine from DEET (what's that?), maybe it is easy to perform, maybe not.
Post 447646 (missing)
(Rhodium: "DEET -> Diethylamine", Newbee Forum)The coupling reaction sounds good though, but then again: where to obtain the coupling reagent from? Synthing your own isn't that easy either (peptide chemistry is a chapter in itself).
That's covered. They can be bought without suspicioun, one needs to spend only 10 minutes to find a suitable supplier for peptide couplers who mail worldwide. Those who seek shall find. PyBOP is easiest to get around here, so one would assume this would be used however SWIM's friend touted using a relatively new coupler designed for coupling dialkyl groups or, meaning a higher yeild, since it's coupling with an dialkylamine.
In regards to using CDI, read this post.
Post 458213 (https://www.thevespiary.org/talk/index.php?topic=12738.msg45821300#msg45821300)
(PapaSmerck: "flipper yield discrepancies", Tryptamine Chemistry)Because of it's byproduct to CO2, basic spikes will probably lower yeilds. SWIM thinks HCTU is a better option. This performed better on tests then BOP/PyBOP, with less racemization which I believe is something we can do without.
Contstructive critisim please!
Post by: armageddon on August 26, 2004, 08:47:00 AM
How does the reactivity of the indole amine compare to the reactivity of diethylamine?
It is by far the weaker base. As long as there is excess diethylamine, the indolic NH2 won't react to any appreciable rate (or did I miss something?)
n00dle:
Ok, so it seems the coupling reagent wouldn't be a big problem, provided one has a rotovap and inert gas setup; this peptide coupling thing sounds more and more interesting to me (especially as I like "toss in and stir for x hours"-reactions very much :) ) - but a problem which arises no matter if CDI, BOP, pyBOP or others are used: how much woodrose seeds would one bee have to extract to get sufficient (lets say 1g) precursor after thorough purification? Multiple kilos? How to obtain such quantities (ok one could buy it - expensive)? And 50 gallons of solvent? (but I agree with you: the product justifies every work and hassle, as long as you don't deal with a serious health hazard like phosgene or cyanogen bromide)
Greetz A
Post by: n00dle on August 26, 2004, 10:03:00 AM
Inert gas setup: argon/nitrogen tank, im sure these can be got from welding, regulators, just make friends with a welder and all your problems are solved. Into a 2-holed stopper in a flask place some glass tubing extending down to the bottom of the flask, and in the other hole, a bit of tube and connect it to a very VERY weak valve so you dont overpressurise enough to blow the vessel. Im talking like, even a water airlock would be sufficient. Charge the vessel with your solvents and chemicals, and very slowly regulate some inert gas through the solvent/chemical solution and up into the flask. This will create positive pressure blowing the air out of the flask, and also by bubbling it through the solution (argon/nitrogen won't react easily at all) you will remove any dissolved oxygen/Co2. After you've flushed the flask (do an oxygen/co2 test at the end of the water-airlock) turn off the gas and leave it connected. Voila, one reaction vessle with no air in it.
A rotovap would be handy, but shouldnt really be necessary, if one chooses the right solvents simply evaporating under vaccum should be fine. But if one insists, there is a ghetto-prep for a rotovap on Rhodium's site.
But a problem which arises no matter if CDI, BOP, pyBOP or others are used: how much woodrose seeds would one bee have to extract to get sufficient (lets say 1g) precursor after thorough purification?
Again on Rhodiums page, 200grams of woodrose seeds would provide roughly 1 gram of LSA which upon hydrolysis using NaOH or KOH 0.5gm of usable lysergic acid. But, SWIM just read about using tetrabutylammonium hydroxide at 30C for 20 hrs to yeild 80% with less than 3% of iso. Figuratively speaking, at less than 3% iso, a person may not even choose to seperate that isomer as it won't do anything anyways, and it's in such small quantity (keep in mind, purity is your god).
200gm of seeds is easy to come by. Even taking say 20 seeds and planting them out, in 2 years you'd have a sustainable source of woodrose seeds. SWIM's friend's 2 year old plant covers a whole side of his house, and regular pods have 4 seeds in them. Plus, they seed twice a year, so one could take multiple harvests and place the seeds into a vaccum bag and suck all the air out, keeping it in a dry dark place should preserve them from degredation until enough seeds can be aquired.
[/blue]And 50 gallons of solvent?
For 200gm seeds, 1liter of nonpolar and 1liter of slightlypolar solvent were used. I think 2L of solvent is jusitified for a person's private lifetime amount of 'cid.
but I agree with you: the product justifies every work and hassle, as long as you don't deal with a serious health hazard
woodrose seeds --> toluene defat --> A/B extract to Lysergic Acid tartarate --> dump in some tetrabutylammonium hydroxide (exotic yes, but worth it, shouldn't be watched) to get 80% usable lysergic acid with a trace of iso-lsd, removed by changing PH and filtering, then reverting to original PH. DEET bug repellent can be bought anywhere, dump that into 10% NaOH re:rhodium's post and distill into hcl, boill off the water to get diethylamine. Dump 1eqmol lysergic acid into 1.05 eqmol PyBOP reagent and 2 eqmol diethylamine, keep it stirring for 3 hours, remove the lsd with a solvent, and chromatograph as freebase or tartarate salt.
Nothing here is really toxic or that dangerous, and because you only really need one chromatograph at the end (presuming you defatted the seeds like a mofo and you acid/base extracted correctly), so almost all of this can be done in the dark or with minimal light and therefore could be done in a day easily. Prepare everything beforehand, and even if you have to bring it into the light for 10 seconds to see what you're doing, big deal, no massive degredation.
Post by: SpicyBrown on August 26, 2004, 04:39:00 PM
It is by far the weaker base. As long as there is excess diethylamine, the indolic NH2 won't react to any appreciable rate (or did I miss something?)
That's what I thought, just wasn't 100% sure. Thanks. Guess that takes care of that portion.
n00dle, that's excellent regarding the amide hydrolysis. With respect to the procedure involving tetrabutylammonium hydroxide - if it is indeed a far superior route, a preliminary SciFinder search seems to indicate that quarternary ammonium hydroxides can be made from their corresponding iodides using silver oxide in water. Around here, tetrabutylammonium iodide is extremely common.
As to the coupling agents, do you (n00dle) have any idea what this new coupler suited for dialkyl groups is? Would be worth looking into. These coupling reactions are nice and fast as well, in solid-phase peptide synthesis someone I once knew used HBTU as the coupling agent, allowing each amino acid to react with the HBTU (DMF as solvent) for 2 minutes prior to addition to the peptide resin, where it was allowed to run for 5 minutes before flushing the reactants away; this minimized racemization and gave high coupled yields in under 10 minutes, which is pretty cool if you think about it. Never used HBTU for solution-phase coupling however; both EDC for normal amide formation and 1-hydroxy-7-azabenzotriazole (HOAt) for dipeptide linkage have been personally observed by my previous friend to produce exceptionally high yields.
The inert gas setup is indeed a breeze; by bargain-hunting, someone put together a setup with a small tank and regulator for under $100, the argon fill was around $15. This is all in the name of welding.
I really think this seems like a worthwhile idea, even worth running two columns for purity's sake. :)
-SpicyBrown
Post by: armageddon on August 27, 2004, 03:40:00 AM
I'm a chemist, no flower grower :) ! And they're harder to harvest than wine (manually of course) - 50 seeds usually amount to 6 grams, so harvesting would take a bit of time even if 4 seeds can be collected at once ::) . But they can also be bought, that's true - and if they indeed contain 0.5% lysergic alkaloids, it is not that expensive anymore. Man, you're lucky! Where SWIA lives, HBWR don't become that old (too cold here), and most surely they don't become 2-yeared, cover whole houses and seed twice per year! Wish I lived where you do (is it maui/hawaii? *lol*)... (SWIA is green with envy :) )
So to return to your original question: what is difficult about making acid? The only possible problems I could think of can easily be circumvented, as you both pointed out. So I myself begin to wonder about the simplicity of using peptide coupling agents, and why it hasn't gained much more popularity!?
(oh, don't forget to use brown glassware or wrap your clear glassware with aluminum foil or similar; lysergic compounds aren't that light sensitive as you might think; better be on the safe side though.
But still: the lighting problem/hype cannot really be the reason for the lack of poluparity with these rxns IMHO!)
A
Post by: n00dle on August 27, 2004, 08:58:00 AM
Post by: hest on August 27, 2004, 11:07:00 AM
The big problem is obtaining the lyserg acid (or some kind of ergotamine).
Personal I would make it with peptidecouplings, but the shulgin way is also fine.
Post by: n00dle on August 27, 2004, 02:56:00 PM
..redundant statement.
The big problem is obtaining the lyserg acid
This was already addressed in the thread. The starting material is not in question. Surely you noticed that when you clicked on the thread called 'LSD VS peptide coupling...'
(or some kind of ergotamine) There are different kinds of the same chemical?
Personal I would make it with peptidecouplings, but the shulgin way is also fine.
Sigh.. Please post things that are relevant to the topic, nothing in your statement actually helped any of us out, it ws merely some form of discussion, and discussions belong in 'The couch' forums. Please redirect statements like your last post there
Post by: armageddon on August 27, 2004, 06:07:00 PM
But true is that the precursor problem is not an issue of this thread anymore.
Sure, the shulgin way is well-explored - but not as nice to work with as BOP/HBTU/DCC (is CDI normally used in the same fashion like these; for coupling amino acids?) from what I can see.
Which peptide coupling reagent would you suggest Hest? And why?
A
Post by: n00dle on August 27, 2004, 06:57:00 PM
Post by: moo on August 27, 2004, 07:10:00 PM
Post 353379 (https://www.thevespiary.org/talk/index.php?topic=12853.msg35337900#msg35337900)
(Rhodium: "New LSD analogs from the Nichols Lab", Tryptamine Chemistry)?Post by: armageddon on August 27, 2004, 07:48:00 PM
pyBOP reacts with the acid to give an acyloxyphosphonium salt and the amine reacts with this intermediate to give the end product.
As a side note, pyBOP is used in place of BOP, since BOP yields the carcinogenic HMPT as a side product.
Those phosphonium coupling reagentia give better yields than DCC (dicyclohexylcarbodiimide).
(
Post 355652 (https://www.thevespiary.org/talk/index.php?topic=12853.msg35565200#msg35565200)
(Cyrax: "Nice, they use the pyBOP coupling reagent, which ...", Tryptamine Chemistry)- seems like it was good to know that before beginning to choose one's coupling reagent!?
And one thing I don't get: in the original Nichols paper Rhodium references to, they use 2,4-dimethylazetidine (secondary amine, too) instead of diethylamine - but what's the role of diisopropyl-ethylamine (tertiary amine)? Is it used to act like a buffering agent? (for buffering what?)
Synthesis of Lysergamides (general method)
Lysergic acid monohydrate (200mg, 0.75mmol), pyBOP (426mg, 0.82mmol), and the appropriate 2,4-dimethylazetidine (109mg, 0.9mmol; replace this with equimolar amount NH(Et)2 to yield lysergic acid diethylamine) were suspended in 20ml of CH2Cl2. Diisopropylethylamine (193mg, 1.5mmol) was added, and the reaction was stirred for 3h. The reaction was then quenched by the addition of 20ml of 0.75 M concentrated NH4OH. The CH2Cl2 layer was separated, and the aequous phase was extracted with 10ml of CH2Cl2. The organic layers were combined and washed with H2O (2x 30ml) and brine (15ml) and dried (MgSO4). Filtration and solvent removal by rotary evaporation under reduced lighting, followed by drying under high vacuum, produced a light golden foam. This crude product was then subjected to purification(..)
..sounds indeed really easy! But why the tertiary amine?
Greetz A
Post by: n00dle on August 28, 2004, 06:08:00 AM
On the sidenote, a total guess, that maybe they add a higher boiling amine to the lower boiling amine that is used to couple, so that the solutions BP is raised. People have had issues trying to couple with some amines that have BP's of 3 degrees celcius. But then you'd wonder, you'd probably get a mixture of coupled lysergic acid amines. Hmm. Either way, as proof of the methods on Rhodium's site, a 'supporting' amine is not required. FANTASTIC! This is excellent news.
(Off Rhodium's page)
Coupling with BOP-Reagent
Example:
A solution of tert-butyloxycarbonyl threonine (2.19g, 10mmol) (the carboxylic acid) and phenylalanine methyl ester hydrochloride (2.16g, 10mmol) (amine hydrochloride) in 150mL ACN is stirred at RT while the BOP-reagent (4.42g, 10mmol) is added, followed by the addition of triethylamine (2.2g, 2.8mL, 20mmol). The rxn is stirred at RT for 1.5hr. 100mL of a saturated NaCl solution is added and the product extracted with EtOAc 3x. The combined organics are washed with 2N HCl, H2O, 5% NaHCO3, and then H2O. The organics are dried over MgSO4, filtered, and concentrated in vacuo to give the dipeptide (3.74g, 98%).
Here they even use the hydrochloride salt of the amine! This helps us also as it is easier to work with diethylamine hydrochloride than diethylamine itself.
WEEEEEEEEEEEE :D
Post by: armageddon on August 28, 2004, 06:54:00 AM
And I just wanted to point out that the synthesis I retyped originates from
https://www.thevespiary.org/rhodium/Rhodium/pdf/azetidine-lsd.pdf (https://www.thevespiary.org/rhodium/Rhodium/pdf/azetidine-lsd.pdf)
, the topic of the thread Moo linked too - so thank him for the insight, and maybe me for retyping.....but I mainly retyped it for getting quicker help with my problem - understanding the role of tertiary amines in this coupling reaction! triethylamine (2.2g, 2.8mL, 20mmol) is used in the general peptide synthesis on Rhod's page you posted here, again there is some tertiary amine present, and I'm not very content until knowing about the purpose of doing so - although the fact that this synth seems to work nicely for making lysergamides is indeed already good to know, just for educayshunnal purrposess ;D ..
(oh, and about But then you'd wonder, you'd probably get a mixture of coupled lysergic acid amines. Hmm. Either way, as proof of the methods on Rhodium's site, a 'supporting' amine is not required. React carboxylic acid with a tertiary amine? How would that work? But anyway, it was just your guess... and I guess you meant the sketchy, old doc on Rhod's site suggesting the use of peptide coupling reagents, not the general peptide coupling reaction with BOP you posted? 'Cuz the procedure in your post uses tertiary amine too, so I would say it is needed with these rxns for some reason?!)
(hey wait! I think I even know a biologist doing lab work regularly, and HIS SPECIALTY ARE AMINO ACIDS :) - maybe I will ask him about the topic, surely would be interesting to learn about from someone with experience in the field... ;) )
;D Greetz A ;D
Post by: n00dle on August 28, 2004, 10:46:00 AM
Yeah, I was shooting shit in the wind about that last statement. However just because a tertiary amine is required doesnt meant one has to understand why! ( althought it'd be nice to know :D ) Perhaps it's used to stop some sort of side reaction or something. But procuring some triethylamine or a tertary amine might be a bit more tricky. Any ideas for this?
Post by: armageddon on August 28, 2004, 06:41:00 PM
Exactly - personally, I would rather try to fully understand the reaction before experimenting with ergot precursors from seeds - simply too much work involved in its isolation, therefore I wouldn't want to fuck up any of it because of my own stupidity - better learn about it before doing it).
After having done the incredible and UTFSEing for tertiary amine :P - some tertiary amine syntheses:
Post 488687 (https://www.thevespiary.org/talk/index.php?topic=11745.msg48868700#msg48868700)
(Organikum: "Mono and trimethylamine by electrolysis", Novel Discourse)Post 374511 (missing)
(terbium: "No gas is evolved.", Chemistry Discourse)Post 450362 (https://www.thevespiary.org/talk/index.php?topic=12648.msg45036200#msg45036200)
(Chimimanie: "Maybee", Tryptamine Chemistry)...and maybe here lies the answer to my question about tertiary amines, too:
Post 400250 (https://www.thevespiary.org/talk/index.php?topic=13159.msg40025000#msg40025000)
(Vibrating_Lights: "Missing part", Tryptamine Chemistry)- May I cite Vibrating_Lights:
"The amidization is run in DCM. 3eq of trimethylamine is added dissolved into the DCM then the Lysergic is added followed by the thionyl Chloride then the Diethylamine. the triethylamine is there to imediatly soak up the HCL as it is formed. otherwise the HCl is going to harm the molocule. The triethylamine is too large of a molocule to add to the Lysergic acid chloride so that is not a concern. Following the rxn the sulution is concentrated in vaccume and is taken up in Alcoholic NH3. This is extracted in ethylacetate and run through a cromotography collum."
and further, about the question if different tertiary amines can be substituted:
"either will work. and yes it can be used with the POcl5 synth. Honestly though if you can get get POCl5 then you can probbly get DCC&HBTU. The DCC route is much cleaner i hear. A good friend routinuely uses dcc&Hbtu in the coupling of amino acids and highly recommends it over any of the other coupling techniques. It is good pratice too when working with LA hydrate to induce the LA into the rxn vessel dissolved in DCM and pass it through a silica drying tube to dry it. these silica tubes should be used in introducing any reactants. this will also keep moisture from the atmosphere from entering. Back to the DCC it is very mild and very fast. with that synth basicly one would just add reactants and in 30 mins it is done and yeilds over 80% with little or no iso-lsd produced. There are refs for all these procedures."
Hmm.. VL: If you read this, would you care to answer my little question? (why is tertiary amine used) and maybe tell us a bit about the use of DCC? That would be nice, too..
...anbd suddenly, the next question arises :) : which solvent to use? Anhydrous conditions, introducing reactands through silica column - or dumping them just into de-ionized ice-water? (I never heard about water being used as solvent for makin 'cid, that's why I'm unsure about this!) Guess it strongly depends on which coupling reagent is used, no?
And, update about required equipment: BIG soxhlet xtractor & condenser! (for seed extraction :) )
Greetz, A
Post by: n00dle on August 29, 2004, 11:54:00 AM
Like I originally said, it's probably there to stop another reaction.
The silica tube is a fantastic idea.
Do not use a soxhlet extractor for this preperation, surely you know lysergic acid diethylamide is severely destroyed by heat, the same should apply to lysergic acid amide. There is nothing wrong with pulverising the seeds, washing in nonpolar toluene or xylene to defat, then washing out with ethanol. Figuring the compound one is trying to achieve, I think from this gunk, a chromatography is in order. But hell, a column doesnt take long to run and it certainly is worth it. SWIM has almost no chromatography experience but surely someone has the right solvent/values already documented for chromatographing lsd. The same solvents can be used for LSA, but i think you'd need a new RF value to calculate the elutent propotions.
Post by: hest on August 29, 2004, 04:06:00 PM
one: after the coupling the reamains of the couplingreagent (ecept dcc) is an acid part therby the base ore your resulting amide would be an salt
Two: some of the reagents tends to produce the anhydride of the base iff the tert.base is not present (and deprot. the acid)
Post by: hest on August 29, 2004, 04:27:00 PM
Acid (3 eq.) dissolved in DMF and Nem (MW 115, 4 eq., r=0.91) and TBTU (MW 321, 3eq.) is added. Wait 5 min, then add amine to the mixture. Reaction time 10min-2h
Coupling with PyBOP
. Martinez et al. J. Med. Chem. 28 pp1874 (1986).
J. Coste et al, Tetrahedron Lett. 31 pp.205 (1990)
3eq.acid, 4 eql PyBOP (520.3 g/mol), and 6 eq DIEA (129ul/mmol) in DMF then add amine reac time 10min-1h
coupling with HATU
L.A. Carpino, J. Am. Chem. Soc. 115 pp. 4397-4398 (1993)
acid (3 eq), HATU (2.8 eq), HOAt (1 eq) and NEM (3 eq) are mixed in DMF and preactivated 1-2 min at R.T. then add amine. reaction time 10-20 min.
Solvents. Usual couplings are done in DMF but that is becourse we are talking solid phase chemistry. When wee use wet chemistry DCM or something els will do the trick, as long as all the componets stay in solution
Post by: armageddon on August 29, 2004, 05:14:00 PM
Do not use a soxhlet extractor for this preperation, surely you know lysergic acid diethylamide is severely destroyed by heat, the same should apply to lysergic acid amide. There is nothing wrong with pulverising the seeds, washing in nonpolar toluene or xylene to defat, then washing out with ethanol
Temperature? Weeeell... LSA isn't that fragile - in plants it survives the summer without problems at least! ;) Maybe use soxhlet under vacuum (lower bp)? Soxhlet is superior because it is much quicker and uses way less solvent than the bucket method.. :)
hest: Thanks a lot!!! :)
..but WTF is
Nem (MW 115, 4 eq., r=0.91)?
and HOAt (1 eq)?
DIEA is diisopropylethylamine, I would guess?
Greetz, A
Post by: hest on August 29, 2004, 05:40:00 PM
http://www.polycarboninds.com/html_pages/activ_for_pep_synth.html (http://www.polycarboninds.com/html_pages/activ_for_pep_synth.html)
HOAt adition might be more important when working with acive esters (the Pfp esters usual used in solid phase chemistry)
HA
Post by: armageddon on August 29, 2004, 10:48:00 PM
...seems like ANY tertiary amine in 3x excess over carboxylic acid should do just fine to soak up any acidic by-products! ;D (but HOAt seems useful with CDI, too)
- now THAT'S good news dare I say!
(beginning to wonder about which coupling reagent should be favoured - think it is pyBOP... :) )
Benzotriazole-1-yl-oxy-tris-pyrrolidino-phosphonium hexafluorophosphate
CAS NO.: 128625-52-5; C18H28N6OP2F6; M.W. 520.3
Solubility: 1 mmole in 2 ml DMF clearly soluble
HPLC: purity: ? 97.00%.
The standard in situ coupling reagent for solid phase peptide synthesis [1-5]. This product can replace BOP in all applications without loss of performance. Unlike uronium-based reagents, PyBOP® does not give rise to guanidinated by-products during cyclization and fragment condensation reactions.
[1] J. Martinez, et al. (1985) J. Med. Chem., 28, 1874.
[2] J. Coste, et al. (1990) Tetrahedron Lett., 31, 205.
[3] G. B. Fields, et al. in "Innovation & Perspectives in Solid Phase Synthesis, 1st International Symposium", R. Epton (Eds), SPCC UK Ltd., Birmingham, 1990, pp. 241.
[4] T. Høeg-Jensen, et al. (1991) Tetrahedron Lett., 32, 7617.
[5] R. von Eggelkraut-Gottanka, et al. (2003) Tetrahedron Lett., 44, 3551.
taken from: NovaBiochem Cat.-Nr. 01-62-0016 (
http://www.merckbiosciences.co.uk/product/01-62-0016 (http://www.merckbiosciences.co.uk/product/01-62-0016)
)So the suggested procedure so far would look like this:
Aquire seeds, crush, dry in dessicator (or similar). Defat by extracting thoroughly with nonpolar (preferably low-boiling, like DCM), remove solvent traces with vacuum, extract *very* thoroughly with lots of dry methanol (maybe using vacuum soxhlet), concentrate in vacuo, then column chromatograph and maybe a/b to isolate LSA, hydrolyze LSA to lysergic acid (KOH).
Separate iso- from lsyergic (HNO3) and isomerize with KOH. Combine the two portions of "normal" (non-iso) lysergic acid, and separate optically pure d-lysergic acid via tartrate. Store cold/dark and under inert gas.
Make diethylamine from DEET bug repellant. (make sure you have very good purity here, too)
Make trimethylamine with one of the known methods (I would go for paraform/NH4Cl/160°C, but that's just me) or aquire any other tertiary amine - Purify *very* thorougly...
Turn off the light - illuminate your lab with candles... :)
Dissolve 1 molar eq. d-lysergic acid tartrate in ice-cold analytical grade DCM (dried over mol. sieves) in brown glass flask under inert gas atmosphere, add 1.1 eq. pyBOP followed by 4 eq. tertiary amine as the free base, stir for five minutes. Then add 2 eq. diethylamine hydrochloride and stir for 3 hours under cooling. When reaction is completed (check with kaiser test), quench with conc. ammonium hydroxide, separate the organic layer, extract 4x with more DCM, combine organics. Wash 2x with ice-cold d.H2O, then 2x with brine (use analytical grade NaCl here, NO TABLE SALT please!) and dry thoroughly with molecular sieves. Place in small evaporation flask (brown glass), rinse sieves 3x with small amount of DCM, and evaporate solvent with a rotovap. Column chromatograph residue to (hopefully) get pure d-lysergic acid diethyl amide.
- any comments, improvements, critique? What could be done different? And any input about the solvent/rf. values to use for chromatographing the starting material amine, as well as the product amide?
Greetz, A
Post by: armageddon on August 30, 2004, 07:36:00 AM
http://www.erowid.org/plants/morning_glory/morning_glory_extraction2.shtml (http://www.erowid.org/plants/morning_glory/morning_glory_extraction2.shtml)
Post 505109 (https://www.thevespiary.org/talk/index.php?topic=5399.msg50510900#msg50510900)
(doktor_alternate: "practicality of large scale column chromatography", Chemicals & Equipment)Post 488767 (https://www.thevespiary.org/talk/index.php?topic=12897.msg48876700#msg48876700)
(Freemings: "LSD chromatographia", Tryptamine Chemistry)Post 482624 (https://www.thevespiary.org/talk/index.php?topic=12948.msg48262400#msg48262400)
(Sleen: "LSD - CDI Method?", Tryptamine Chemistry) (this one answers the chromatography question quite well)Post 436693 (https://www.thevespiary.org/talk/index.php?topic=12977.msg43669300#msg43669300)
(chemotype: "hmmm... 250 mg of ergotamine tartrate", Tryptamine Chemistry) (this is even better :) )--
and a very sad thread, too:
Post 350684 (missing)
(Rhodium: "Half-a-Pint - RIP", General Discourse)(May you have a happy lab time there, Halfapint! The HIVE collective will never forget you.)
A
Post by: hest on August 30, 2004, 09:40:00 AM
N-( (R )-l-Methylpropyl)-9,lO-didehydro-6-methylergoline-
B&carboxamide (2). (+)-Lysergic acid monohydrate
(150 mg, 0.52 mmol) and 25 mL of dry, ethanol-free CHCl, were
placed in a flamedried 50 d, three-necked, round-bottom flask
equipped with a condenser, N2 line, and septa inlets. The stirred
slurry was brought to reflux in a preheated 90 "C oil bath after
which 384 mg (5.2 mmol) of (R)-(-)-2-butylamine (Aldrich) in 1.0
simultaneously, via syringe, over 3 min. The mixture was allowed
to stir at reflux for an additional 5 min and was then cooled to
room temperature. The clear amber CHC13 solution was then
washed with 1 M NHdOH (3 x 30 mL) and brine (1 X 10 mL)
and dried (Na,SO,). The drying agent was removed by fitration,
and the solution was concentrated in the dark by rotary evaporation
at 30 "C.
The residue was purXed and fractionated by radial centrifugal
chromatography (Chromatotron, Harrison Research) using a silica
gel rotor and eluting with ethyl acetate in an N2-ammonia atmosphere.
TLC (silica gel, EtOAeNH3) showed a large blue
fluorescent product spot at R, 0.21 corresponding to the (R)-2-
butyllprgamide and a much smaller spot at Rf 0.15 corresponding
to the S isomer. The faster moving component was collected and
concentrated by rotary evaporation. The residue was taken up
into CH2ClZ, washed with HzO, and dried (MgSOJ, and the
CHzClz was removed by rotary evaporation followed by pumping
under high vacuum. The free base (158 mg, 94% yield) was taken
up into 2 mL of methanol, and 57 mg of maleic acid in 0.75 mL
of methanol was added. The maleate salt (1:l stoichiometry)
spontaneously crystallized as a white crystalline solid mp 210
"C dec; lH NMR (free base, CDClJ S 0.91 (t, J = 7.5 Hz, 3 H),
1.13 (d, J = 6.7 Hz, 3 H), 1.36-1.56 (m, 2 H), 2.60 (s,3 H), 2.68-2.82
(m, 2 H), 3.05-3.13 (m, 1 H), 3.31-3.56 (m, 3 H), 3.89-3.98 (m,
(m, 3 H), 8.15 (br 8, 1 H); [(Y]D = +48" (c = 0.1, HZO); IR (free
base, neat) 1650 cm-l (C=O). Anal. (CzrH&I3O5) C, H, N.
N - (( S ) - 1 -Met hylpropyl)-g, 1 0-didehydro-6-met hylergolina88-
carboxamide (3). An exact replication of the above
procedure using 150 mg of (+)-lysergic acid monohydrate and 384
mg of (S)-(+)-2-butylamine (Aldrich) gave 154 mg (91.7% yield)
of the free base. The free base in methanol was combined with
55 mg of maleic acid in methanol to again yield a white aytalline
solid with 1:l stoichiometry: mp 213 "C dec; 'H NMR (free base,
CDC13) 6 0.93 (t, J = 7.5 Hz, 3 H), 1.13 (d, J = 6.7 Hz, 3 H),
1.37-1.58 (m, 2 H), 2.60 (a, 3 H), 2.66-2.82 (m, 2 H), 3.05-3.13
(m, 1 H), 3.31-3.56 (m, 3 H), 3.89-3.98 (m, 1 H), 6.45 (e, 1 H),
6.60 (d, J = 8.0 Hz, 1 H), 6.92 (8, 1 H), 7.12-7.23 (m, 3 H), 8.02
(br 8, 1 H); [(Y]D = +59" (c = 0.1, HzO); IR (free base, neat) 1650
cm-l ( C 4 ) . Anal. (CurH~N305) C, H, N.
This is amides of lysergacid made by the old synthesis by Nichols. look at the yeald's >90% on a sub g. scale.
Read also J.Med.Chem 1995,38,958-966 here Nicholas et.al produse amides from racemic amines and then purify them on their rotochrome (just a fancy prep. TLC plate). Yeald in the 90's (high 40's of each isomer)
Post by: n00dle on August 30, 2004, 10:56:00 AM
Aquire seeds, crush, dry in dessicator (or similar). Defat by extracting thoroughly with nonpolar (preferably low-boiling, like DCM), remove solvent traces with vacuum, extract *very* thoroughly with lots of dry methanol (maybe using vacuum soxhlet), concentrate in vacuo, then column chromatograph and maybe a/b to isolate LSA, hydrolyze LSA to lysergic acid (KOH).
if you chromatograph it it will be pure, no need to a/b extract it. a/b extraction should be done before chromatograph, only if the quantity of extract is quite large.
Separate iso- from lsyergic (HNO3) and isomerize with KOH.
Use tetrabutylammonium hydroxide with or without NaOH to get 3-2.8-0% iso-lysergic acid plus this hydroxide yeilds 30% yeilds. Precipitate the trace isomers and throw them out (not enough to bother hydrolysing). This will save lots of time doing multiple hydrolysis.
Combine the two portions of "normal" (non-iso) lysergic acid
(wont be necesarry if one uses tetrabutylammonium hydroxide)
and separate optically pure d-lysergic acid via tartrate.
Seperate/store as the maleic salt as it has been shown that lysergic acid stored as the maleate salt is even more stable than tartarate.
Store cold/dark and under inert gas.
Make diethylamine from DEET bug repellant. (make sure you have very good purity here, too)
Make trimethylamine with one of the known methods (I would go for paraform/NH4Cl/160°C, but that's just me) or aquire any other tertiary amine - Purify *very* thorougly... ( Electrolysis looks good here. Keep the monomethylamine for other things ;) )
Turn off the light - illuminate your lab with candles...smile (yes i was thinking that, since candles dont put off UV or put off very little?
Your prep was good but there were lots of little things, like the order that would be better, so i re-wrote it.
Get your lysergic acid from the step before, still as base, and chuck it straight into this already-prepared reaction system, there is no point storing it as lysergic acid tartarate. Plus, it needs to be freebased for coupling, so you'd have to spend -more- time freebasing it gently. Anyways, since the PyBOP is the main reaction mechanism, this is to be added last, of the rxn: 4eq triethylamine, 2eq diethylamine hcl, 1eq lysergic acid in minimal NP solvent, and 1.05PyBOP.
SWIM is considering a syringe rxn. Suck up the triethylamine first, then lysergic acid tartarate in d.h2o, then diethylamine hcl in d.h2o, then pybop in water (hoping it's soluble in water). Take the syringe, dump it in your fridge and don't open it for a few hours. This might save people the hassle of creating an inert atmosphere. Boiled water shouldn't contain much dissolved gasses. Just fill any leftover space in the syringe with d.h2o and you should have an environment that is inert enough for the 1-2 hour reaction.
Chromatograph with hexane/ethyl acetate in 5% fractions. Still not sure which of these fractions to collect. I guess evaporate them all and find the one that has the most solid product, this is probably the 'peak' of the spectrum containing the most of the lsd product.
Novel idea: When you have chromatographed your LSD, mix it with some free ascorbic acid to form Lysergic Acid Diethylamide Ascorbate. It has been shown in psylocibin that as the ascorbate salt, was stable at room temperature and light exposure and did not readily oxidise to psilocin. Since ascorbic acid is an antioxidant, and LSD is not stored in light normally, the main amount of oxidation will be from oxygen in the air. It is SWIMs oppinion that since psilocybin ascorbate did not oxidise at RT/light, that LSD ascorbate will probably be even more resistant to oxidation than lsd tartarate or lsd maleate. (Comments plz?)
Post by: hest on August 30, 2004, 12:20:00 PM
EXTENSIONS AND COMMENTARY : LSD is an unusually fragile molecule and some comments are in order as to its stability and storage. As a salt, in water, cold, and free from air and light exposure, it is stable indefinitely. There are two sensitive aspects of its structure. The position of the carboxamide attachment, the 8-position, is affected by basic, or high pH, conditions. Through a process called epimerization, this position can scramble, producing isolysergic acid diethylamide, or iso-LSD. This product is biologically inactive, and represents a loss of a proportionate amount of active product. A second and separate point of instability is the double bond that lies between this 8-position and the aromatic ring. Water or alcohol can add to this site, especially in the presence of light (sunlight with its ultraviolet energy is notoriously bad) to form a product that has been called lumi-LSD, which is totally inactive in man. Oh yes, and often overlooked, there may be only an infinitesimal amount of chlorine in treated tap water, but then there is only an infinitesimal amount of LSD in a typical LSD solution. And since chlorine will destroy LSD on contact, the dissolving of LSD in tap water is not appropriate.
I Think that Shulgin has a good point here. The big LSD-killer is not light but chlorine ions.
And for the reaction 1: use DCM or DMF as solvent. 2: Don't use the Hydrochloride of DEA use the freebase.
3 seperate on an prep.TLC plate, up to 1g can bee splited at once, an you don't have to learn collumchrom first.
And n00dle could you give me the ref for the tetrabutylammonium hydroxide hydrolysation, I would love to read it
Post by: n00dle on August 30, 2004, 02:56:00 PM
../rhodium/chemistry
/paspalic.lysergic.html (https://www.thevespiary.org/rhodium/Rhodium/chemistry
/paspalic.lysergic.html)
Post by: armageddon on August 30, 2004, 04:07:00 PM
You don't need tor freebase it when you use the tetiary amine as a free base - it will do this in situ if enough excess tert. amine is present - same thing like with diethylamine*HCl. But as the neutralization produces heat: maybe it would really be better to work only with free bases and forget about in-situ basification..
About the order of reactands: "pyBOP reacts with the acid to give an acyloxyphosphonium salt and the amine reacts with this intermediate to give the end product." - as I stated before.
So maybe it would be better to first mix lysergic acid with pyBOP and tetiary amine buffer, let stir for x minutes to form the acyloxy salt, and then add diethylamine to be coupled with and stir for 2-3h.
About the candles emitting almost no UV light: yes, that was my thought behind it, too - and the total amount of light is also very small, although they still allow for enough lighting to be able to work with... not to forget the mystic atmosphere... :)
Chromatograph with hexane/ethyl acetate in 5% fractions. Still not sure which of these fractions to collect. I guess evaporate them all and find the one that has the most solid product, this is probably the 'peak' of the spectrum containing the most of the lsd product.
Maybe one could check with a small UV light and collect the fluorescent band/spot/fraction when chromatographing 'cid..
And:
Post 415643 (https://www.thevespiary.org/talk/index.php?topic=12738.msg41564300#msg41564300)
(Lilienthal: "LSD is not that light sensitive as you might think", Tryptamine Chemistry)"Table II illustrates that fluorescent light can cause decommposition of LSD in transparent containers when they are placed in close proximity [15 cm] to the light source. Under these conditions, the half-life of LSD was approximately 4 weeks. As the distance between the source of fluorescent light and the samples increased, the percent of LSD decomposition decreased. The results demonstrate that LSD can withstand normal room light conditions at a constant temperature of 25°C for 1 week without noticeable structural change."
:)
Greetz, A
Post by: hest on August 30, 2004, 05:18:00 PM
So maybe it would be better to first mix lysergic acid with pyBOP and tetiary amine buffer, let stir for x minutes to form the acyloxy salt, and then add diethylamine to be coupled with and stir for 2-3h.
Yes that is eksatly how you perform an usual peptide couling.
Post by: n00dle on August 30, 2004, 06:09:00 PM
Lysergic acid MW 268.315
Diethylamine MW = 73.14
Triethylamine MW 101.2
Lysergic Acid Amide MW = 269.315
Tetrabutylammonium Hydroxide MW = 259.4744
NaOH MW = 39.99707
Based on these MW's that SWIM found, and based on using 1gram of peptide coupler (was most expensive chemical in rxn) the following numbers were formed, could someone plz check them.
Hydrolysis
1.5eq TBAOH x 1eq NaOH x 1eq purified woodrose alkaloids
0.003405mol Tetrabutylammonium hydroxide : 0.00227mol NaOH : 0.00227mol purified woodrose alkaloids
0.8835gm Tetrabutylammonium hydroxide : 0.09079gm NaOH : 0.6125gm purified woodrose alkaloids
Rxn = 80% efficiency, Yeilds 0.49gm Lysergic Acid.
Coupling
1.05eq peptide coupler : 1eq lysergic acid : 2eq diethylamine : 4eq triethylamine
0.00192mol pybop to 0.001828mol lysergic acid to 0.003656mol diethylamine to 0.007312mol triethylamine
1 gram pyBOP : 0.49gm lysergic acid : 0.2674gm diethylamine : 0.7399gm triethylamine
rxn efficiency unknown, but assumed high 90's.
Yeilds lysergic acid diethylamide.
Post by: armageddon on August 30, 2004, 07:02:00 PM
And about the price of pyBOP: I've seen 5 grams for 110 bucks - think the woodrose thingie would be the more expensive part, at least if your house isn't incidentally covered with them... ;)
Everything else seems right so far.
Greetz A
Post by: n00dle on August 30, 2004, 09:01:00 PM
The tetrabutylammonium hydroxide gave hydrolysis yeilds of 80% with 3% iso-lysergic acid or less.
Using tetrabutylammonium hydroxide requires a little under 0.7gm of total alkaloids. 200gm of seeds yeilds roughly 1gm, so about 150gm-180gm of seeds would be required for a 450mg lsd freebase synthesis. Commercially this would be expensive, but most ethnobotanists will have a woodrose plant, and since LSA is not the most inviting chemical to partake with, would probably have lots of seeds to sell/get rid of to a person at very discounted prices. (They would otherwise be wasted.)
Post by: armageddon on August 30, 2004, 10:40:00 PM
What about d/l rotation? lysergic acid cannot only isomerize (iso/normal) but also has a optical rotation (d/l). Only the d-lysergic acid gives LSD-25. That's why I mention the tartrate purification all the time: it allows for separating d- from l-lysergic acid. Isomerization and rotation are not the same thing. And if a synthesis would start with a mixture of d/l lysergic acid, the product would partially consist of inactive l-lysergic aicd diethylamide and therefore, dosaging the product would be a wild guess. But if the synth starts with optically pure d-lys, the end product will have same optical purity.
So go get some l-tartaric acid - it is not only used for storage! ;)
Greetz A
Post by: n00dle on August 31, 2004, 01:08:00 AM
Post by: armageddon on August 31, 2004, 01:42:00 AM
But I'm not very sure about that; maybe some more experienced bee could help us with that ;) ?
Another instruction on how to chromatograph LSD-25:
"A solid residue of 3.45 gm. comprising the "normal" and "iso" forms of d-lysergic acid N,N-diethylamide is obtained. This material is dissolved in 160 ml. of a 3-to-1 mixture of benzene and chloroform, and is chromatographed over 240 g. of basic alumia. As the chromatogram is developed with the same solvent, two blue fluroescing zones appear on the alumina column. The more rapidly moving zone is d-lysergic acid N,N-diethylamide which is eluted with about 3000 ml. of the same solvent as above, the course of the elution being followed by watching the downward movement of the more rapidly moving blue fluorescing zone. The eluate is treated with tartaric acid to form the acid tartrate of d-lysergic acid N,N-diethyl amide which is isolated. The acid tartrate of d-lysergic acid N,N-diethyl amide melts with decomposition at about 190-196 degrees centigrade.
The di-iso-lysergic acid N,N-diethyl amide which remains absorbed on the alumia column as the second fluroescent zone is removed from the column by elution with chloroform. The "iso" form of the amide is recovered by evaporating the chloroform eluate to dryness in vacuo."
(
../rhodium/chemistry
/lsdpatent.html (https://www.thevespiary.org/rhodium/Rhodium/chemistry
/lsdpatent.html)
Greetz A
Post by: n00dle on August 31, 2004, 02:21:00 AM
Post by: armageddon on August 31, 2004, 04:21:00 AM
Maybe some bee in the know could help here?
About your first post in this thread: It's too easy.. - there you have your complications. :P Not as easy as it looked at first sight. But we'll figure it out, would I say! :)
Greetz A
Post by: hest on August 31, 2004, 01:25:00 PM
From the fermentation of Claviceps you only get's the right isomer (usual you get's all kind's of ergo..wathewerer), but after the hydrolyse you have the right isomer of the accid.
PErsonal I think this is the easyest way to 1g of the acid, but that is another (alredy cowered)
Post 519261 (https://www.thevespiary.org/talk/index.php?topic=13024.msg51926100#msg51926100)
(Bubbleplate: "Process for Isolation of Indole Alkaloids", Tryptamine Chemistry) threadPost by: n00dle on September 01, 2004, 11:42:00 AM
Unless of course hydrolysis step yeilds different optical isomers. If it doesnt, then there's nothing to worry about.
Plus even if one doesnt seperate any theoretical optical isomers or even isomers, the amounts should be so small and won't use up any signifigant amount of coupling reagent. Plus, forming the tartarate salt at the end and chromatographing should leave both iso-lsd and the useless optical isomer behind, no?
In regards to extracting them, why can't we reuse the solvent by means of acid/base extraction? Or maybe a ghetto watercooled soxhlet with a low BP solvent would help.
Post by: armageddon on September 01, 2004, 05:19:00 PM
Chromatography separates iso- from normal, but not d- from l-lysergic acid diethylamide. But fractional crystallization via l-tatrate gives optically pure products (d-/l-..)
I just thought that using a racemic mixture of d/l-lysergic in the coupling step would mean a racemic end product - and this would mean half of the coupling reagent got used up forming the unwanted l-lysergic acid DEA ( :) ), therefore it might bee advantageous to separate d- and l- before coupling it with the secondary amine.
But I just thought about ephedra: this plant produces only one optical isomer, too - chances are good that HBWR will do, too... :-[
What's ghetto about soxhlet with methanol? :) The soxhlet apparatus would have to be water-cooled anyway (better said the condenser it is equipped with), and when slight vacuum is applied, the necessary heavy reflux can be had at slightly above room temp. - doubt that this will destroy any LSA, as the temp. of the boiling solvent would be <50°C (depending on vacuum strength)...
(and LSA is not as fragile as LSD-25)
greetz A
Post by: armageddon on September 01, 2004, 05:42:00 PM
The residue was purXed and fractionated by radial centrifugal
chromatography (Chromatotron, Harrison Research) using a silica
gel rotor and eluting with ethyl acetate in an N2-ammonia atmosphere.
TLC (silica gel, EtOAeNH3) showed a large blue
fluorescent product spot at R, 0.21 corresponding to the (R)-2-
butyllprgamide and a much smaller spot at Rf 0.15 corresponding
to the S isomer. The faster moving component was collected and
concentrated by rotary evaporation (they separate R/S isomers)
Ehmmm... - now I'm very puzzled: they separate R/S via chromatography?? I thought R/S was the same like d/l, whereas the iso/normal orientation was molecular, not optical? :-[
Strange, especially when seen in connection with the following (taken from other paper):
"As the chromatogram is developed with the same solvent, two blue fluroescing zones appear on the alumina column. The more rapidly moving zone is d-lysergic acid N,N-diethylamide" (...) "The di-iso-lysergic acid N,N-diethyl amide which remains absorbed on the alumia column as the second fluroescent zone is removed from the column by elution with chloroform. The "iso" form of the amide is recovered by evaporating the chloroform eluate to dryness in vacuo." (they separate iso- from normal LSD)
Now what does column chromatography do exactly? separating iso from normal, d- from l- (or R- from S-), both..?
(Help! I can't believe that noone of the >500 bees having already visited this thread does know an answer to this!?)
Greetz, A
Post by: hest on September 01, 2004, 09:13:00 PM
There are two types of steric compounds
Isomers that are mirror images of each other are caled enantiomers, just like d and l amphetamine. They have only one chiral carbon atom.They are both chirale and have identical properties in an symmetrical (non chirale) enviroment, that mean the same meltingpoint ect.
This mean that they are imposible to separate on a standard TLC plate, iff you want to separate them you need an chiral TLC plate.
The next type are diastereomers these molecules have more than one optical carbon and have different chemical propertiesthat means different meltingpoints ect and, you can seperate them on TLC plates.
So you can separate your mixture of four types of lysergacid into two mixtures of racemic mixtures.
So to anser the quistion we have to know witch type you want to separate.
The short anser is that they separate iso from the fun stuff. They start wit the right isomer of lyserg acid (as you will doo)
Post by: Rhodium on September 01, 2004, 09:23:00 PM
Hest: I assume that you are not talking about chloride ions here, but rather free chlorine and any charged species formed by its aqueous dissolution? I don't think chlorine can be formed to any great extent by UV irradiation of aqueous chloride solutions, or the ocean surface would be sterile.
Post by: armageddon on September 01, 2004, 09:46:00 PM
About the stereoisomers: So there are d-isolysergic, l-isolysergic, d-lysergic and l-lysergic.
What we want is every "d-" form, so assuming that HBWR don't contain only d-LSA but also l-LSA (I still don't believe plant alkaloids are optically pure :) ), tartrate salt formation via l-tartaric would be in order first (and maybe some l-(iso)-lysergic would have to be discarded - or can it be converted to something useful?), followed by separation of "iso" from "normal" (by column chromatography or via pH adjustment/extraction) then isomerization of d-iso-lys. into d-lysergic (or use the LSA->lysergic hydrolysis method suggested by nOOdle, since it produces nearly no iso), and then one would have pure d-lysergic acid diethylamide with very little iso contamination, upon pyBOP coupling w/(Et)2NH, right?
(always assuming the LSA->lysergic acid hydrolysis step would give a racemic mixture of all four diastereomers)
BTW Thanks a lot for clarifying on that! 8)
Greetz A
Post by: armageddon on September 01, 2004, 10:12:00 PM
Aquire seeds, crush, dry in dessicator (or similar). Defat by extracting thoroughly with nonpolar (preferably low-boiling, like DCM), remove solvent traces with vacuum, extract *very* thoroughly with lots of dry methanol (maybe using vacuum soxhlet), concentrate in vacuo, then column chromatograph and maybe a/b to isolate LSA, hydrolyze LSA to lysergic acid (KOH).
Separate iso- from lsyergic (HNO3) and isomerize with KOH. Combine the two portions of "normal" (non-iso) lysergic acid, and separate optically pure d-lysergic acid via tartrate. Store cold/dark and under inert gas.
Make diethylamine from DEET bug repellant. (make sure you have very good purity here, too)
Make trimethylamine with one of the known methods (I would go for paraform/NH4Cl/160°C, but that's just me) or aquire any other tertiary amine - Purify *very* thorougly...
Turn off the light - illuminate your lab with candles...
...should do quite well! (only correction: first a/b on plant extract, then column chromatography) :)
Greetz A
Post by: trade_om on September 02, 2004, 02:12:00 PM
Post by: armageddon on September 02, 2004, 04:31:00 PM
From "Merck Index":
"D-Tartaric acid: (2S,3S)-2,3-Dihydroxybutanedioic acid
(unusual tartaric acid; unnatural tartaric acid; l-tartaric acid; (-)-tartaric acid; levotartaric acid; D-threo-2,3-dihydroxysuccinic acid)
Levorotatory tartaric acid having a dextro configuration.
(2R,3R)-2,3-Dihydroxybutanedioic acid
(ordinary tartaric acid; natural tartaric acid; d-tartaric acid; (+)-tartaric acid; dextrotartaric acid; L-2,3-dihydroxybutanedioic acid; d-a,b-dihydroxysuccinic acid; Weinsäure (German); Weinsteinsäure (German))
Dextrorotatory tartaric acid having a levo configuration."
About optical resolution with D-(-)-tartaric acid:
Post 511856 (https://www.thevespiary.org/talk/index.php?topic=7748.msg51185600#msg51185600)
(Rhodium: "the freebase should be used", Stimulants)Post 506241 (https://www.thevespiary.org/talk/index.php?topic=13322.msg50624100#msg50624100)
(Rhodium: "chirality of otc tartrate salts", Tryptamine Chemistry)Post 474569 (https://www.thevespiary.org/talk/index.php?topic=12689.msg47456900#msg47456900)
(bbell: "Hawian baby woodrose", Tryptamine Chemistry)Post 417270 (https://www.thevespiary.org/talk/index.php?topic=12757.msg41727000#msg41727000)
(pHarmacist: "Sandoz Ltd. LSD synthesis (1946)", Tryptamine Chemistry)Post 275468 (https://www.thevespiary.org/talk/index.php?topic=12767.msg27546800#msg27546800)
(El_Zorro: "Re: trouble getting yields for LSD", Tryptamine Chemistry)I recommend this one - so I will retype it: :)
"This base was dissolved in warm, dry MeOH, using 4 mL per g of product. There was then added dry d-tartaric acid (0.232 g per g of LSD base), and the clear warm solution treated with Et2O dropwise until the cloudiness did not dispel on continued stirring. This opaqueness set to a fine crystalline suspension (this is achieved more quickly with seeding) and the solution allowed to crystallize overnight in the refrigerator. Ambient light should be severely restricted during these procedures. The product was removed by filtration, washed sparingly with cold methanol, with a cold 1:1 MeOH/Et2O mixture, and then dried to constant weight. The white crystalline product was lysergic acid diethylamide tartrate with two molecules of methanol of crystallization, with a mp of about 200 °C with decomposition, and weighed 3.11 g (66%). Repeated recrystallizations from methanol produced a product that became progressively less soluble, and eventually virtually insoluble, as the purity increased. A totally pure salt, when dry and when shaken in the dark, will emit small flashes of white light.
(taken from the LSD-25 entry in TiHKAL...)
And some fun stuff on the topic:
Post 477135 (https://www.thevespiary.org/talk/index.php?topic=13011.msg47713500#msg47713500)
(Mr_Rodgers: "sensitivity of ergolines", Tryptamine Chemistry)Post 481216 (https://www.thevespiary.org/talk/index.php?topic=12739.msg48121600#msg48121600)
(Rhodium: "An Interview With an LSD Clandestine Chemist", Tryptamine Chemistry)(took 2 minutes of UTFSE ;) )
Question to those in the know: is it really D-(-)-tartaric acid which results in crystallization of d-isomers while leaving l- in solution? I begin to wonder because the common naming id D- and L-, but Merck sez that d-tartaric is in fact L-tartaric - I would guess that shulgin referred to D-(-)-tartaric?
Greetz, A
Post by: n00dle on September 03, 2004, 01:08:00 AM
No chromatography was used on the woodrose extract, or on the end product, but lysergic acid dimethylamine fine pointy crystals were obtained. WEEEE!
Writeup coming in future.
Legal: Yes, diMETHYLamine was used as lysergic acid diethylamide is a scheduled compound.
Also, about the potassium metal in ether/methanol reflux to isomerize, is that 98% or so yeilds, what's the percentage of lefover iso's? Because, refluxing lysergic acid amide with K to 98%, then precipitating the iso and using K again to get 98% recovery, is like, a 96% overall yeild. Couple that with the high yeilding peptide coupling methods and WHOA, we've got one VERY VERY efficient rxn for what used to be a very difficult and intricate operation.
Post by: armageddon on September 03, 2004, 01:51:00 AM
But don't bee too sure about the legality of LS-dimethylamide..
And another thing google revealed..
About ergot alkaloid content of ipomoea/rivea seeds:
"In 1955 Osmond described personal experiments with Rivea corymbosa seeds and reported that the effects were similar to those of d-lysergic acid diethylamide (LSD-25). He suggested (1957) that the word psychedelic (meaning mind-manifesting) be used as a generic term for this class of substances to refer to their consciousness-expanding and psychotherapeutic function as contrasted with the hallucinogenic aspect. In 1960 Hoffman reported that he had isolated d-lysergic acid amide (LA) and d-isolysergic acid amide from the seed of both Rivea corymbosa and Ipomoea violacea."
I guess Hofmann would've reported the discovery of l-stereomers too. Hm - anyway, reXtalization via d-tartaric is also a good purification method...
Greetz A
Post by: n00dle on September 03, 2004, 02:24:00 AM
A simple acid base extraction of woodrose alkaoids and rextallisation gave satisfying crystals.
or does it maybe even emit sparkles when shaken (autoluminescence)?
It was not pure so piezoluminescense wasn't all that great BUT... (see next post)
Did your cat/dog/whatever get high on it?? wink
..The trustable person who performed this rxn says this product is definately active.
But don't bee too sure about the legality of LS-dimethylamide..
It could probably be marked under the analogue act but as far as solid law goes, lysergic acid diethylamide is the only solidly scheduled item. But either way, this is why i wouldn't attempt this rxn, because of the legal reasons. Though if SWIM worked in a proper lab where lysergamide synthesis was needed, BOY OH BOY would this be fun.
Lol, imagine getting a job at a chem synthesis company, and using these techniques. I think the boss would be a bit confused when you got outstanding yeilds, better than thier prepatory methods :P
n00d.
Post by: armageddon on September 03, 2004, 02:35:00 AM
It was not pure so piezoluminescense wasn't all that great BUT... (see next post)
- can't await it! PLZ hurry up, can't wait to see... ;)
I think the boss would be a bit confused when you got outstanding yeilds, better than thier prepatory methods.
Probably - especially when you come up with a lot of hive-powered special procedures to achieve you high yield (how high was it BTW?) - I namely think of "diethylamine from DEET bug repellant"... ;D
(no joke: he would probably be quite impressed and/or ashamed!?)
about the potassium in ether/methanol hydrolysis (do you have a ref for this, too?):
Because, refluxing lysergic acid amide with K to 98%, then precipitating the iso and using K again to get 98% recovery, is like, a 96% overall yeild
I guess you meant hydrolysis of the amine to the carboxylic acid, yielding 98% d-LS? Then the remainder would be d-iso-LS, I would assume - that is 2%. If you really want to bother with that: 98 percent (of starting material) plus (0.98 * 2) percent of the starting material would be 99.96 percent yield of d-lysergic acid from LSA in two steps - assuming that there are no losses during rxn or isolation, which is of course NOT true. So I would just stick with the 98% and call that a very good yield of pure d-lysergic for a one-step hydrolysis... ;)
Greetz, A
Post by: n00dle on September 03, 2004, 03:02:00 AM
The synthesizer will synthesise it again im sure, as soon as he/she is informed of the potassium reflux, i'll make sure she/he informs us of the PyBOP coupling, however this time SWIM has a feeling he'll use PyBrOP (the coupler specifically for dialkyls?) so hopefully there should be an even higher yeild for this. ;D ;D ;D
Post by: armageddon on September 03, 2004, 05:34:00 AM
- but we know that KOH reflux isomerizes at the position characterized as "iso" or "normal" and not at the chiral "d/l"-center of the molecule - at least it is the standard route to change "iso-" to "normal" LS compounds, besides LSA->LS hydrolysis.
It results in d-LS* as well as a smaller amount of d-iso-LS*, but no l-(iso)-LS* - which is maybe produced by some coupling reaction, unless pyBOP or similar is used...
(I hope this was understandable :) )
So I would think that potassium metal would work much in the same way, but somewhat more thoroughly (i.e. 85% normal/iso vs. 70%(?) normal/iso).
But as said, I'm not sure about whether overall yield or yield of the desired isomer is meant - but the better yield means more of the desired isomer anyway, right? (or would you think it maybe yields 98% of a 70/30 iso/normal mixture? that would be baaad, since after 3 isomerization steps, almost everything would've been converted to iso...) ;D
How was the yield BTW?
Greetz
Post by: n00dle on September 03, 2004, 05:09:00 PM
Still trying to get that writeup, person is unavailable at the moment, but it will be posted here as soon as it is recieved.
Post by: n00dle on September 03, 2004, 05:24:00 PM
Jordi Alsina, George Barany, Fernando Albericio, and Steven A. Kates
Lett. Pept. Sci. 6, 243-245 (1999).
"Pyrrolidide derivatives are observed as unwanted by-products from slow reactions of activated carboxylates with nucleophilic amines, as mediated by phosphonium salt coupling reagents (PyAOP, PyBOP, PyBroP). This side reaction is attributed to the presence of small amounts (e.g., 0.5%, w/w) of pyrrolidine as a contaminant to commercial phosphonium salts, and does not occur when the reagents are crystallized before their use in coupling reactions."
Lysergic acid pyrrolidide is pretty darn impotent compared potency of lsd (1:20th the potency, on rough average), so it is recommended to rextallize the coupler before use.
Post by: n00dle on September 03, 2004, 05:30:00 PM
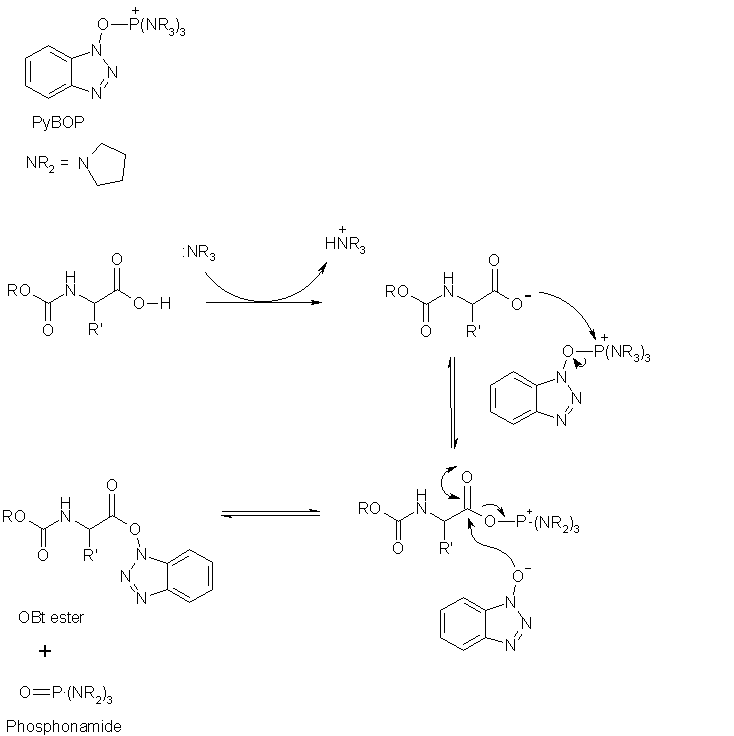
Hope this answers why tertiary amines are needed!
Post by: armageddon on September 03, 2004, 06:40:00 PM
How was the yield the experimenter was able to obtain?
Greetz
Post by: n00dle on September 03, 2004, 07:48:00 PM
But it does work, so SWIM guesses we will just have to wait for another peroson (or him/her) to run the rxn again.
Post by: armageddon on September 10, 2004, 05:32:00 PM
"Thin layer chromatography was carried out on purchased aluminium roll, Silica gel F254 (E. Merck AG, Darmstadt, Germany). The Rf values for LSD, iso-LSD and lysergic acid in the solvent system, chloroform:methanol (3:2, v/v) are 0.74, 0.45 and 0.06 respectively. The spots were detected by fluorescence under UV light 254 nm) and by spraying with 1% N,N-dimethylaminobenzaldehyde in ethanol:HCl(aq) soln. (sp. gr. 1.18) (1:1, v/v)."
..taken from
../rhodium/chemistry
/14c-lsd.html (https://www.thevespiary.org/rhodium/Rhodium/chemistry
/14c-lsd.html)
.
Post by: chemotype on September 11, 2004, 11:06:00 PM
Post by: n00dle on September 12, 2004, 02:23:00 PM
So? What exactly is your point here? There is a huge dosage change upon lysergic acid amide and lysergic dialkylamides. The dosage for woodrose seed ingestion is anywhere from 4 to 10 seeds. Theoretically, if 4 to 10 seeds were extracted crudely, and hydrolysed, then coupled, the existing -single- dosage of LSA would be transformed into probably 20 LSD dosages. Ingesting 50-100mcg of LSA probably wouldnt do anything or bare effects, but 50-100mcg of LSD certainly will!
Post by: chemotype on September 13, 2004, 05:01:00 AM
Post by: n00dle on September 14, 2004, 08:21:00 AM
Why are you so suprised to see xtals come out of woodrose? Its just an acid/base alkaloid extraction. The better your defatting procedure, the better luck you will have at rextalising. Just like alkaloid extractions from like, any plant.
Post by: meme on October 06, 2004, 03:11:00 PM
The LSAs were very soluble in water, and, although this extraction was not complete it was very clean.
I do not remember whether this specified HBWS, MG, or either seed, though ;) I'd imagine that it is true for either.