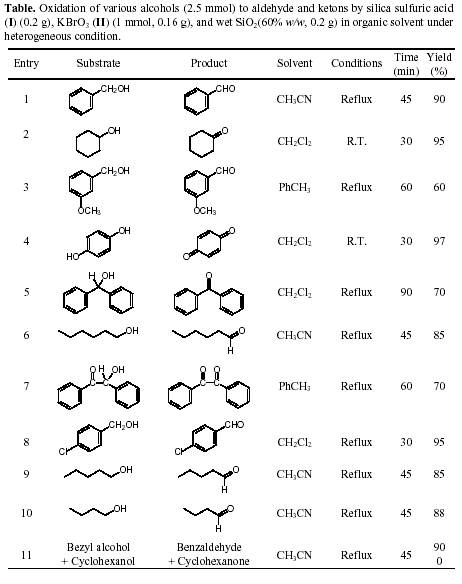
Silica Sulfuric Acid/KbrO3/Wet SiO2 as an Efficient Heterogenous System for the Oxidation of Alcohols Under Mild ConditionsActa Chim. Slov. 2003, 50, 563-568
(
http://acta.chem-soc.si/50/50-3-563.pdf)
Abstract: A combination of silica sulfuric acid and potassium bromate in the presence of wet SiO
2 were used as an effective oxidizing agent for the oxidation of alcohols to their corresponding aldehyde or ketone derivatives in dichloromethane, acetonitrile or toluene in good yields.
ExperimentalGeneral: Chemicals such as alcohols, potassium bromate, dichloromethane, toluene, acetonitrile and silica gel were purchased from Fluka, Merck and Aldrich chemical companies. Silica sulfuric acid was synthesized according to our previously reported procedure.
9-11 The oxidation products were characterized by comparison of their spectral (IR,
1H NMR), TLC and physical data with the authentic samples.
Oxidation of Hydroquinone to p-Benoquinone. A Typical Procedure: A mixture of hydroquinone (0.275g, 2.5 mmol), potassium bromate (0.167g, 1 mmol), silica sulfuric acid (0.2g) and wet SiO
2 (60% w/w, 0.2g) in dichloromethane (5 mL) was stirred at room temperature for 30 minutes. The heterogeneous mixture was filtered and washed with Na
2S
2O
3 solution (10%, 2×20 mL). The organic phase was collected. Anhydrous Na
2SO
4 (2g) was added to the solution and filtered after 15 minutes. Dichloromethane was removed. The solid was crystallized from n-hexane. The p-benzoquinone was obtained in good yield (Table).
 Preparation of Silica Sulfuric AcidSynlett 2003, 1877-1879
Preparation of Silica Sulfuric AcidSynlett 2003, 1877-1879DOI:
10.1055/s-2003-41469
A 500 mL suction flask was used. It was equipped with a constant-pressure dropping funnel containing chlorosulfonic acid (23.3g, 0.2 mol) and gas inlet tube for conducting HCl gas over an adsorbing solution i. e. H
2O. Into it were charged 60.0g of silica gel. Chlorosulfonic acid was added dropwise over a period of 30 min at r.t. HCl gas evolved from the reaction vessel immediately. After the addition was complete the mixture was shaken for 30 min. A white solid (silica sulfuric acid) 76.0g was obtained.