also is there a way to tell if something is soluble in something else by looking at the atomic wieght or boiling temp or anything other than just trying it.You cannot know
for sure until you've tried it, but with increasing experience you learn how to judge the
approximate polarity of a molecule from its structure (basically by comparing the number of polar functional groups with the overall size of the molecule). The presence of charged or ionizable functional groups, such as -COOH, -NH
2 and -SO
3H tend to make substances polar and hence more water-soluble - this at the same time as they become less soluble in non-polar solvents (like toluene). The presence of aromatic rings and carbon chains [-CH
2-]
n tend to make substances insoluble in water and more soluble in non-polar solvents. Another good rule-of-thumb is that the larger a molecule is, the lower will its overall solubility in
any solvent be. Small molecules are often freely soluble in (miscible with) medium-polarity solvents like acetone, ethyl acetate and chloroform (these are especially powerful solvents, as their intermediate polarity allows them to dissolve both substances which are more polar as well as less polar than themselves).
What makes something polar or non-polar?The fundamental reason behind the emergence of the phenomenon, its definition, or its practical implications?
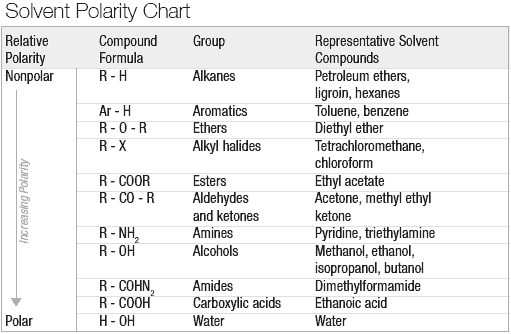
Above you have a polarity chart covering most common common classes of compounds. For the precise polarity index of many common solvents, and a table of which solvents are miscible with each other, see the PDF linked from
Post 343523 (missing)
(Rhodium: "Solvent Miscibility Chart", Newbee Forum)For further information about the practical merits of choosing solvents of varying polarity, see
Post 453146 (missing)
(Rhodium: "Factors influencing choice of solvent", Newbee Forum) The very phenomenon which is responsible for making compounds polar or non-polar is if the electron distribution in the molecule is even or not. As you know, electrons carry negative charge, so if one end of a molecule hogs an excess of the electrons in the molecule, it becomes polar due to its uneven distribution of charge. Different atoms have different tendencies to hog electrons (this is called "electronegativity"), so depending on which atoms are part of a molecule, and how they are arranged in space compared to each other, it will become more or less polar.
The technicalities behind this polarization of bonds and molecules can be read here:
Polar Bonds and Electronegativity
(
http://www.ucdsb.on.ca/tiss/stretton/chem2/arch13.htm)
Molecular Shapes and the Polarity of Molecules
(
http://www.ucdsb.on.ca/tiss/stretton/chem2/arch14.htm)
Solvents are generally categorized into three main types:
1. polar protic (miscible with water, contains O-H bonds)
Ex: Methanol, Ethanol, Isopropanol, Acetic Acid2. dipolar aprotic (miscible with water, but lacks O-H bonds)
Ex: Acetone, DMSO, Acetonitrile, Dimethylformamide (DMF), HMPA3. non-polar (immiscible with water)
Ex: Toluene, Xylene, Dichloromethane, Diethyl Ether, Ethyl AcetateThis is discussed in detail, with many examples,
here
(
http://www.usm.maine.edu/~newton/Chy251_253/Lectures/Solvents/Solvents.html)
Influence of solvent polarity on reaction rate:
Solvent Effects on Reaction Rates - Comparisons
(
http://www.chem.ucalgary.ca/courses/351/Carey5th/Ch08/ch8-8.html)
None
(
http://www-scf.usc.edu/~chem322a/ppt/Chapter_06/322a_Ch_6_18.pdf)
None
(
http://www-scf.usc.edu/~chem322a/ppt/Chapter_06/322a_Ch_6_19.pdf)
Solvent Effects on reaction rates - Solvation
(
http://chemweb.stanford.edu/winter2003/Chem33/Handouts/lecture18b_ho.pdf)