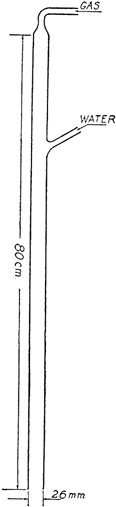
Gas Absorption TrapOrganic Syntheses, CV 2, p. 3A convenient trap devised by John R. Johnson for the absorption of hydrogen chloride, or for the elimination of sulfur dioxide, hydrogen cyanide, etc., may be arranged as shown in the figure.
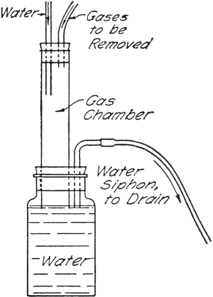
The gases are led into a chamber in which a stream of water (from the reflux condenser in this case) flows downward into a large bottle. The bottle is provided with a bent tube which serves as a siphon drain. The gases are thus brought into contact with a flowing stream of water so that the heat of solution is dissipated, and the level of the water in the lower bottle serves as a seal to prevent escape of the gases into the atmosphere. Water-insoluble gases are drawn out through the siphon drain directly into the sink.
If the gas chamber is of sufficient capacity there is practically no danger of water being drawn back into the reaction vessel, but care should be exercised when the reaction flask is cooled. For the reaction described here, a gas chamber about 2.5 cm. in diameter and 20–25 cm. in length was found to be satisfactory.
In reactions involving compounds sensitive to water it is desirable to introduce a drying tube between the condenser and the trap in order to absorb any moisture which might diffuse up from the trap.
An alternative, and simpler, gas absorption trap is described in
Organic Syntheses, CV 1, p 95: