I've decided to put the pedal to the metal and synth. this MF. I've decided to react Grignard complex with (S)-proline ethyl ester:
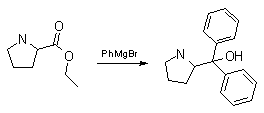
However, I have only proline so I'll have to synth the Et-ester of it mytself.
I have no refs on the esterification part of this particular amino acid but I intend to apply what I know in general. This what I have written in my lab-record-book and what I'm about to follow:
Procedure:Nearly equimolar amounts of (S)-proline and EtOH are placed in a 500 mL RB flask (slight excess of EtOH, I want proline to be limiting since EtOH is wiledly available

. Concentrated H2SO4 is added to the reaction mixture. Mixture is heated under re-flux for about 3 h (to be certain, I have no TLC plates at the time, thus I can not develop TLC).
When 3 h has passed the reaction flask is cooled and the reaction mixture is poured over crushed ice (in a baker). This is stirred around with a glas rod for about a minute or two and transfered to a 200 mL sep. funnel. To minimize losses and to get better yield both reaction flask and ice-baker are rinsed with ether and the washing poured into the sep. funnel. Aqueouse phase run off the drain, and the org. phase is washed with freshly prepared FeSO4 solution and then with 2 times with sodium carbonate solution. Ether phase is dried over MgSO4, filtered and the solvent is striped off under vacuuo. For the sake of quality and pride the residue (crude product) should be distilled once more.
What do you think about the above?

I'm more interested in what you think about my new DEA-HAT!