Here's CA vol. 79, 1973
108478g 108478g New type of Urshibara Nickel catalyst (U-Ni-N). Activation of the precipitated nickel (precursor of Urushibara nickel catalysts) by refluxing with alcohols. Kajitani, Masatsugu; Okada, Junko; Ueda, Tetsuya; Sugimore, Akira; Urushibara, Yoshiyuki (Fac. Sci. Technol., Sophia Univ., Tokyo, Japan).
Chem. Lett. 1973, (

, 777-80 (Eng). A new method is described for activating pptd. Ni by refluxing with alcs. The pptd. Ni activated in this manner exhibits a catalytic activity corresponding to acid-activated pptd. Ni. The catalyst is useful for the selective hydrogenation of 1-octene (low isomerization) and cycloolefins.
And CA Vol. 95, 1981
95:96939y and
95:96940s 95:96939y Optimization of Urushibara nickel catalysts for hydrogenation. Part I. Effect of preparation conditions on the spectific activity of the catalyst. Maksimova, N. A. ; Fasman, A. B. (Inst. Org. Katal. Elektrokhim., Alma-Acta, USSR).
Izv. Akad. Nauk Kaz. SSR, Ser. Khim. 1981, (3), 26-31 (Russ). A math. model was derived to describe the activity of Urushibara Ni catalysts in hydrogenation of HC
÷CCMe
2OH as a function of the prepn. conditions. Improved catalysts were obtained by increasing the Zn content, the concn. of the HCl used to treat the Zn before the onset of cementation, and the cementation temp., and by decreasing the Al content and AcOH concn.
95:96940s Optimization of Urushibara-type nickel catalysts for hydrogenation. Part II. Effects of preparation conditions on the selectivity of hydrogenation of dimethylethynylcarbinol. Fasman, A. B.; Maksimova, N. A.; Pel'menshtein, B. Ya. (Inst. Org. Katal. Elektrokhim., Alma-Acta, USSR).
Izv. Akad. Nauk Kaz. SSR, Ser. Khim. 1981, (3), 31-4 (Russ).
A simplified math. model was derived to describe the effects of the conditions of the prepn. of Urushibara Ni catalysts on the selectivity of HC
÷CCMe
2OH hydrogenation with them. The max. selectivity for CH
2CHCMe
2OH was 93% , obtained using 20% Pd
2+, 40% Mn
2+ and 1:10
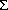
(Ni, Pd, Mn)/Al at pH 6.5, cementation at 40º for 30 min, leaching at 40º with 20% NaOH for 20 min, Al particle size 0.2-0.4 mm, and wash-water temp. 20º.
Note:
÷ means triple bond here.
Where did I get the
from?
http://www.geocities.com/dritte123/PSPF.html