I have this thought about the synthesis of alpha-substituted amines. You might know of some that are interesting.
My thought is this: If there were an easy way to oxidize the amino functionality to a nitro group, the alpha-alkylation could proceed via a nitroaldol (Henry) route, followed by acid-catalyzed dehydration of the resulting alcohol, and then simultaneous reduction of the double bond and of the nitro group back to an amino.
An example might be:
Step 1: Tryptamine --(unknown process)--> Nitro analogue
Step 2: Nitro analogue --------(LDA)--------> Nitro analogue enolate
Step 3: Nitro analogue enolate ---(acetaldehyde)----> Beta-hydroxy alpha-ethyl nitro analogue (BHAENA)
Step 4: BHAENA ----(excess acid)----> Alpha-ethenyl nitro analogue (AENA)
Step 5: AENA -----(H2, Pd)--------> Alpha-ethyl tryptamine
That was a lot of terminology which I'm not sure I'm using correctly. Pictures follow:
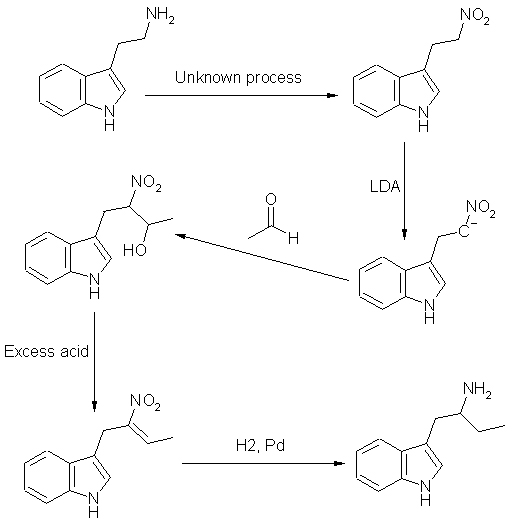
Hopefully what I'm proposing is now clear enough. Questions for discussion:
1. Does anybody know how to do the amino-to-nitro oxidation bit?
2. Does anybody see any major problems with the rest of the plan?