Indium as a Reducing Agent: Reduction of OximesJustin R. Harrison, Christopher J. Moody, Michael R. PittsSynlett (11), 1601-1602 (2000)Abstract?-Oximino carbonyl compounds can be selectively reduced to the corresponding N-acetyl amine with indium powder, acetic anhydride and acetic acid in THF.
The reduction of oximes and oxime ethers to amines is a useful transformation in organic synthesis, and has been achieved using a range of reagents. The most commonly used conditions include: catalytic hydrogenation,
1-3 lithium aluminium hydride,
4 sodium borohydride in the presence of various additives,
5-7 diborane
8 and zinc and acetic acid, often in the presence of acetic anhydride to effect in situ acetylation of the amine.
9-12 In continuation of our interest in oxime chemistry,
13 we needed to reduce the ketoxime 1b to the corresponding N-acetylamine 2b. Attempts to perform this transformation by catalytic hydrogenation over Pd/C with in situ acetylation with acetic anhydride proved unsatisfactory. Thus, we explored the possibility of extending our new indium metal reduction methodologies for the reduction of oximes.
The potential of indium metal as a reducing agent has only recently been recognised, and is based on its first ionisation potential (5.8 eV),
14 which is lower than reducing metals such as zinc (9.4 eV) or tin (7.3 eV) and close to that of strongly reducing alkali metals such as sodium (5.1 eV). In this context we have reported the selective reduction of aromatic nitro groups,
15 the heterocyclic ring in quinolines, isoquinolines and quinoxalines,
16 and the deprotection of 4-nitrobenzyl ethers and esters with indium metal.
17 Subsequently others have shown that indium can be used in the reductive elimination of 1,2-dibromo compounds,
18 and in the reduction of ?-halocarbonyl compounds and benzyl halides,
19 azides,
20 and amine N-oxides.
21 We now report a new method for reduction of oximes to N-acetyl amines using indium metal in the presence of acetic anhydride and acetic acid in THF.
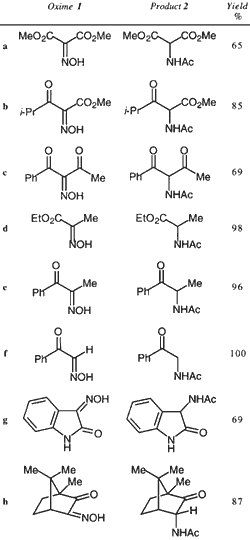
The reduction was carried out by simply heating the ?-oximino carbonyl compound 1 (commercially available or prepared by standard methods) in THF containing 4 equivalents of acetic acid and 2.5 equivalents of acetic anhydride, and a suspension of 4 equivalents of indium powder.
22 The reactions were complete in less than 30 min for compounds in which the C=N bond is rendered more reactive by the presence of two carbonyl groups, and within 18 h for monocarbonyl compounds, and gave the required N-acetyl amines 2 in good to excellent yield (Table).
23 Hence the reaction can be used to prepare a range of ?-acetamido carbonyl compounds including a diester 2a, ?-ketoester 2b, ?-diketone 2c, ester 2d, ketones 2e, 2f, 2h, and amides 2g; the camphor derived oxime 1h gave a single endo-diastereomer acetamide 2h. It is worth noting that the carbonyl group in all cases was unaffected (reduction with hydride transfer reagents would yield the ?-hydroxy amine). The reaction is simple to carry out; there is no need for exclusion of air or the use of dry solvents, and the reaction is worked up by simply quenching with bicarbonate and extraction into organic solvent. In most cases further purification by column chromatography was unnecessary.
In summary, the present work extends the use of indium metal as a reducing agent in organic chemistry, and provides a simple method for the reduction of oximes with in situ acetylation of the resulting amine.
Table Reduction of oximes 1 to N-acetyl amines 2 using indium metal-acetic anhydride-acetic acid in THF
 References and Notes
References and Notes(1) Boye, O.; Brossi, A.; Yeh, H. J. C.; Hamel, E.; Wegrzynski, B.; Toome, V. Can. J. Chem. 1992, 70, 1237-1249.
(2) Cipolla, L.; Lay, L.; Nicotra, F.; Panza, L.; Russo, G. J. Chem. Soc., Chem. Commun. 1995, 1993-1994.
(3) Booker-Milburn, K. I.; Dunkin, I. R.; Kelly, F. C.; Khalaf, A. I.; Learmonth, D. A.; Proctor, G. R.; Scopes, D. I. C. J. Chem. Soc., Perkin Trans. 1 1997, 3261-3273.
(4) Rahman, M. A.; Fraser-Reid, B. J. Am. Chem. Soc. 1985, 107, 5576-5578.
(5) Lee, G. S.; Kammermeier, T.; Kaiser, A.; Eibler, E.; Wiegrebe, W. Arch. Pharm. 1991, 324, 177-181.
(6) Demir, A. S.; Tanyeli, C.; Sesenoglu, O.; Demiq, S.; Evin, O. Tetrahedron Lett. 1996, 37, 407-410.
(7) Baruah, B.; Dutta, M. P.; Boruah, A.; Prajapati, D.; Sandhu, J. S. Synlett 1999, 409-410.
(

Feuer, H.; Braunstein, D. M. J. Org. Chem. 1969, 34, 1817-1821.
(9) Zambito, A. J.; Howe, E. E. Org. Synth. 1973, Coll. Vol. 5, 373-375.
(10) Girard, A.; Greck, C.; Ferroud, D.; Genet, J. P. Tetrahedron Lett. 1996, 37, 7967-7970.
(11) Matuszczak, B. J. Prakt. Chem. 1998, 340, 20-25.
(12) Nudelman, A.; Nudelman, A. Synthesis 1999, 568-570.
(13) Hunt, J. C. A.; Lloyd, C.; Moody, C. J.; Slawin, A. M. Z.; Takle, A. K. J. Chem. Soc., Perkin Trans. 1 1999, 3443-3454, and earlier papers in the series.
(14) Cintas, P. Synlett 1995, 1087-1096.
(15) Moody, C. J.; Pitts, M. R. Synlett 1998, 1028.
(16) Moody, C. J.; Pitts, M. R. Synlett 1998, 1029-1030.
(17) Moody, C. J.; Pitts, M. R. Synlett 1999, 1575-1576.
(18) Ranu, B. C.; Guchhait, S. K.; Sarkar, A. J. Chem. Soc. Chem. Commun. 1998, 2113-2114.
(19) Ranu, B. C.; Dutta, P.; Sarkar, A. J. Chem. Soc., Perkin Trans. 1 1999, 1139-1140.
(20) Reddy, G. V.; Rao, G. V.; Iyengar, D. S. Tetrahedron Lett. 1999, 40, 3937-3938.
(21) Yadav, J. S.; Reddy, B. V. S.; Reddy, M. M. Tetrahedron Lett. 2000, 41, 2663-2665.
(22) General Procedure. Indium powder (-100 mesh, 0.92 g, 8.0 mmol) was added to a solution of the oxime 1 (2.0 mmol), acetic anhydride (0.51 g, ~0.47 mL, 5.0 mmol) and acetic acid (0.48 g, ~0.46 mL, 8.0 mmol) in THF (10 mL) and the mixture heated at reflux for 18 h. Saturated aqueous sodium bicarbonate solution (10 mL) was carefully added to the cooled reaction mixture, and stirring continued for 1 h. The resulting mixture was diluted with water (30 mL) and extracted with ethyl acetate (3 x 15 mL). The combined organic layers were dried (MgSO4) and concentrated in vacuo. If necessary, the crude material was purified by flash column chromatography (silica eluting with ~20% ethyl acetate in dichloromethane) to give the N-acetyl amine 2.
(23) 2a, mp 131-132°C (lit.@24 mp 130-132°C); 2b, mp 38-39°C (lit.@25 no mp given); 2c, oil, 2d, mp 37°C (lit.@26 mp 34-35°C); 2e, mp 89-90°C (lit.@27 mp 87-89°C); 2f, mp 83°C (lit.@28 mp 83-84°C); 2g, mp 246-248°C (lit.@29 248-249.5°C); 2h, mp 115°C (from ether) [...] In the reduction of 1b and 1e, the yields obtained with indium were comparable with those obtained using zinc metal under similar conditions.
(24) L'Esperance, R. P.; Ford, T. M.; Jones, M. J. Am. Chem. Soc. 1988, 110, 209-213.
(25) Kuwano, R.; Okuda, S.; Ito, Y. J. Org. Chem. 1998, 63, 3499-3503.
(26) Lee, J. C.; Oh, Y. S.; Cho, S. H.; Lee, J. I. Org. Prep. Proc. Int. 1996, 28, 480-483.
(27) Koga, K.; Yamada, S.-I. Chem. Pharm. Bull. 1972, 20, 539-542.
(28) Yoon, U. C.; Cho, S. J.; Lee, Y.-J.; Mancheno, M. J.; Mariano, P. S. J. Org. Chem. 1995, 60, 2353-2360.
(29) Endler, A. S.; Becker, E. I. J. Am. Chem. Soc. 1955, 77, 6608-6610.