Vacuum steam distillation of P2P - A theoretical enquiryIn order to fully understand the influence of pressure on the steam distillation phenomena I decided to play a little with the Claussius-Clapeyron's equation:
ln(P
2/P
1)=(1/T
1 - 1/T
2)*deltaH/R
I used approximate data and some idealizations to calculate the evaporation enthalpy (deltaH) and the phase diagram of P2P. Data used: b.p. at 1atm: 215°C ; b.p. at 14torr: 100.5°C. The deltaH for water was taken from literature (40660 J/mol). The enthalpy was idealized to bee non-temperature dependent (off course, this is not true).
Results:deltaH(P2P) = 52.86 kJ/mol = 393,87 kJ/kg
Table of calculations:
| temperature | vapor pressures | P[kPa] | mixture boils ata: | steam distillate | composition |
| T[°C] | H2O | P2P | [kPa] | Xb [mol(P2P)/mol] | Wc [g(P2P)/g] |
| 0 | 0.832 | 0.004 | 0.835 | 0.4% | 3.1% |
| 10 | 1.566 | 0.008 | 1.574 | 0.5% | 3.7% |
| 20 | 2.825 | 0.017 | 2.842 | 0.6% | 4.4% |
| 30 | 4.900 | 0.036 | 4.936 | 0.7% | 5.1% |
| 40 | 8.207 | 0.070 | 8.276 | 0.8% | 5.9% |
| 50 | 13.313 | 0.130 | 13.443 | 1.0% | 6.8% |
| 60 | 20.976 | 0.236 | 21.212 | 1.1% | 7.7% |
| 70 | 32.187 | 0.411 | 32.598 | 1.3% | 8.7% |
| 80 | 48.206 | 0.695 | 48.901 | 1.4% | 9.7% |
| 90 | 70.608 | 1.141 | 71.749 | 1.6% | 10.7% |
| 100 | 101.325 | 1.825 | 103.150 | 1.8% | 11.8% |
| 110 | 142.689 | 2.848 | 145.537 | 2.0% | 12.9% |
Error: Table contains the text "" between [tr] and the next [td] markup tag in the table row "[tr]>[td]temperature[/td][td]vapor pressures[/td][td]P[kPa] [/td][td]mixture boils at<sup>a</sup>:[/td][td]steam distillate [/td][td]composition[/td][/tr]".
Error: Table contains the text "" between [/td] and the next [/tr] markup tag in the table row "[tr][td]T[°C][/td][td]H2O[/td][td]P2P[/td][td][kPa][/td][td]X<sup>b</sup> [mol(P2P)/mol][/td][td]W<sup>c</sup> [g(P2P)/g][/td][/tr]".
a It is assumed that the vapor pressure of a two phase system like H2O/P2P equals the sum of each phase partial pressures at the given T. Effects of impurities is ignored.
b X is the molar fraction of P2P in the distillate and if ideal gas assumption is used it equals P(P2P)/P.
c Since P2P density is 1.003 g/ml (Fluka catalog) the w/w% showed in the table can also be taken to equal vol% of the P2P phase in the distillate. Therefore if W=22% a 1dl of distillate will be composed of 22ml P2P and 78ml of water. Off course, this is only in theory.
Phase diagram:
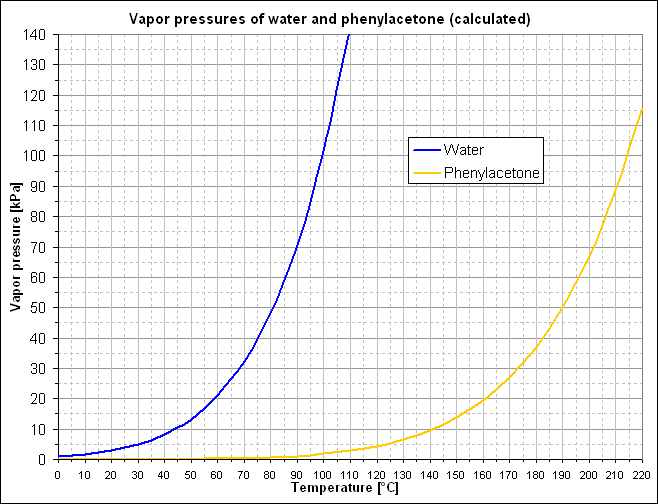
Conclusion
Doing the steam distillation of P2P in a reduced pressure does have some influence on the composition of the distillate. Doing a steam distillation at 40°C (~8.3kPa) causes the distillate to contain two times less P2P (from 12.9% to 5.9%). However, due to the faster process and working at a lower temperature, it might bee practical if a compromise is done. Steam distillation between 60 to 80°C might be faster while still reducing the P2P/H2O ratio of the distillate in an acceptable range.
This conclusion is not to bee generalized to all steam distillable compounds since the results are (very!) strongly dependent on evaporation deltaH which is a specific property of each compound. Small differences in deltaH cause huge variations in the steam distillation. More specifically:
- if the molar deltaH is higher than that of the water, vacuum steam distillation causes a lowering of the non-polar fraction content in the distillate;
- if the molar deltaH is lower than that of the water, vacuum steam distillation causes a lowering of the water fraction content in the distillate;
- if deltaH are equal or very similar, no considerable effect should bee noted.
PS: If someone needs a theoretical phase diagram for any other liquid I can easily do some (now that I have the Excel template). All I need is two b.p. of the compound at two different pressures (normal b.p. and a reduced pressure b.p.).
The post has been heavily edited to incorporate the changes brought by the exact b.p./P data supplied by Armageddon in the next post in this thread. New phase diagram and huge differences in the colclusions! Sorry for the inconvenience.