Please excuse, A lot of my post will be repeat material / (plagiarised from other posts). Also not all of it has to do with the main topic of the thread. However in this post there are structures of heterocyclic stimulants that relate to this thread a greater or lesser degree (e.g. Phacetoperane). It might be useful to have the diagrams of various non-conventional stimulants in one place (along with a brief description), and be able to spin them skeletons around with the mouse (unless the structures crash the molecule viewer and I have to post a flat picture instead!). Also I think there are a couple structures here that weren't yet mentioned too much elsewhere.
Molecule:
diphenyl(piperidin-2-yl)methanol ("OC(C2=CC=CC=C2)(C3=CC=CC=C3)C1NCCCC1")
aka Pipradol, aka Meratran. The "parent" compound. This is legally restricted in sports competition and some countries for modest abuse potential. Sold in tonic.
"Marketed in 1956. Considered to be a mild central nervous system stimulant. Occasionally overstimulation reported. Has been studied in depressive mood. Dosage 1 - 3mg/day."
http://home.intekom.com/pharm/adcock/alerton.html
http://www.psychotropics.dk/alphaindex/view_Alpha.asp?Startchar=M
The compound without the alcohol group, 2-diphenylmethyl piperidine, has also been mentioned in the paper: "in doses of 1 mg/kg s.c. produces a strong increase of the spontaneous motility of animals. On mice it has about the same potency as methamphetamine and is about 3 times more potent than amphetamine but has practically no peripheral sympathomimetic effects". Also pointed out in 'Future Drugs of Abuse': "A best bet for a future CsA is the most potent adrenomimetic compound in this series, 2-diphenylmethylpiperidine."
Molecule:
(R)-(+)-alpha,alpha-Diphenyl-2-pyrrolidinemethanol ("O[C@@](C2=CC=CC=C2)(C3=CC=CC=C3)[C@@H]1NCCC1")
This is obviously a close analog of pipradol. One Hive post said "5-10 mg lasts hours, but not fun", another said "20 mg of the (R) isomer (orally) provides quite a mild, but prolonged stimulation (12 hours) which is quite similar to about 30-40mg of methylphenidate... 20mg IM is 1.5 to 2x oral potency, but it's a lot more euphoric. It also has very little peripheral actions (no heart pounding out of your chest & very little dry mouth)." Apparently works by Dopamine Reuptake Inhibition, like cocaine.
DAT uptake inhibition: Ki is 170 nM vs Cocaine 200 nM
Molecule:
(4-(1,2-diphenylethyl)pyridine ("C1(CC(C3=CC=CC=C3)C2=CC=NC=C2)=CC=CC=C1")
Another similar structure; This was from the thread containing
Post 475625 (missing)
(silenziox: "18 & 23 Cas numbers???", General Discourse)Another Monoamine Reuptake Inhibitor; it has test tube potency comparable to cocaine (but not aware of any bioassays):
DAT Uptake Inhibition: Ki is 263 nM; 5HT Uptake Inhibition: Ki is 910 nM; NE Uptake Inhibition: Ki is 393 nM
Molecule:
2-benzhydryl-3-methoxy-1-methylpiperidine ("COC1C(C(C3=CC=CC=C3)C2=CC=CC=C2)N(C)CCC1")
Substituted 2-diphenylmethylpiperidine has similar potency: "Minimal cardiac and respiratory effects at stimulant doses and was tried clinically. It was a potent stimulant, but the dose-response effect was too marked. At a given dose, due to the variation in the individual patient response, the level of stimulation attained frequently was above or below that desired." Stimulant at 1 mg/kg ip. N-desmethyl analog: Stimulant at 0.3 mg/kg"
Molecule:
alpha,alpha-diphenyl-3-morpholinemethane ("C1(C(C3=CC=CC=C3)C2=CC=CC=C2)COCCN1")
Fastandbulbous states it's a stimulant (DAT inhibitor, as is methylphenidate and cocaine), but it was found to be active at a much lower dose than the other two, (reckoned to be 0.5 - 1mg in man). Lillienthal listed the reference for the synthesis on his references page:
Central Stimulants. Cyclized DiphenylisopropylaminesStanley O. Winthrop and Leslie G. Humber,
J. Org. Chem. 26( , 2834-2836 (1961)
, 2834-2836 (1961)
Molecule:
3-methyl-2-phenylmorpholine ("CC(NCCO2)C2C1=CC=CC=C1")
Phenmetrazine, aka Preludin. A post on the Hive says "Apparently you have to inject phenmetrazine to feel it's true euphoria... When you do that it apparently kicks amphetamine's ass. It was abused much in Sweden in the 60-70s". Oral dosage 50mg 2-3 times a day. Another post states "stimulate the release of Norepinephine like U4Euh does; notice the structural similarity also". Paper says "One widely used approach for the synthesis of new compounds that possess some type of central nervous system stimulant activity is the cyclization of substituted phenethanolamines into heterocycles, such as morpholine, piperidine, and 2-oxazoline in such a manner that more or less of the phenethanolamine skeleton becomes part of the heterocyclic ring. Well-known drugs of this type are phenmetrazine. pipradol, and anminorex."
Post 429511 (missing)
(alphacentauri: "Phenmetrazine", Stimulants)
Molecule:
Phenyl-2-piperidylmethanol ("OC(C2=CC=CC=C2)C1NCCCC1")
aka Phacetoperane: antidepressant, anorexic agent. "Shulgin (1975) suggested that levophacetoperane could well be a future clandestine CsA. However, this compound shares the same limitation as methylphenidate, in that only one diastereoisomer is active; and it is less potent than methylphenidate" Again has the phenethanolamine in heterocycle.
Molecule:
1-(1-benzylbutyl)pyrrolidine ("CCCC(N2CCCC2)CC1=CC=CC=C1")
"Prolintane hydrochloride is a mild central stimulant and has properties similar to those of dexamphetamine. Peak plasma level 1 to 2 hours after single dose. Elimination half-life about 2 hours." There is also the closely related Pyrovalerone:
Molecule:
4'-methyl-2-(1-pyrrolidinyl)valerophenone ("O=C(C1=CC=C(C)C=C1)C(N2CCCC2)CCC")
"Pyrovalerone, Studied as psychostimulating agent in the management of asthenic and depressive states with mental exhaustion." These two above (prolintane and pyrovalerone) could be called the "parent" molecules for the pyrrolidinopropiophenones posted above, synthesized by Nemo Tenetur.
Molecule:
4-(4-chlorophenyl)-1-methyl-3-propylpiperidine ("ClC1=CC=C(C2CCN(C)CC2CCC)C=C1")
This molecule could be considered a piperidine based cocaine analog (or more closely, phenyltropane analog) It is mentioned in
https://www.thevespiary.org/rhodium/Rhodium/pdf/cocaineanalogs.pdf
, page 968. It looks simple and potent (about 30x cocaine in the test tube). It has a reasonable uptake/binding ratio ("agonist"), and as a bonus both stereoisomers have significant activity.
Binding to DAT (cis isomer): Ki 3.0 nM; DAT Inhibition (cis isomer): Ki 8.3 nM
Molecule:
(S)-methyl 2-(3,4-dichlorophenyl)-2-((S)-piperidin-2-yl) ("O=[C@](OC)[C@H]([C@@H]2CCCCN2)C1=CC(Cl)=C(Cl)C=C1")
Here is a halogenated Methylphenidate analog with greater potency than the parent compound (mentioned in the same review paper as the piperidine derivative above.)
DAT Binding: Ki 5.3 nM; DAT Inhibition: Ki 7.0 nM
Molecule:
3-(3,4-dichlorophenyl)-N-methyl-1-indanamine ("CNC1CC(C3=CC=C(Cl)C(Cl)=C3)C2=C1C=CC=C2")
aka Indatraline, Lu 19-005
According to preliminary investigations, potential antidepressant agent. Very long-lasting and potent cocaine-like stimulant, self administered in monkeys but with long intervals in between, because of long duration of action. Produces stereotypies, weight loss, anemia similar to those caused by chronic high-dose cocaine. Combines an equally potent inhibitive effect on dopamine, norepinephrine and serotonin.
Molecule:
4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-7-methoxy-2-methylisoquinoline ("CN2CC1=CC(OC)=CC=C1C(C3=CC(Cl)=C(Cl)C=C3)C2")
aka Diclofensine.
NE/5-HT/DA uptake inhibitor. Psychostimulating and mood elevating agent. Double-blind clinical trials in the treatment of depressions and dysphoric mood. Minor anticholinergic effects. Dosage 25 to 150mg/day. Plasma half-life in healthy subjects about 4 hours. Related to Nomifensine, which itself had recreational and motivating potential and which many people liked before it was removed from the pharmaceutical market.
1-(3,4-dichlorophenyl)-3-azabicyclo-[3.1.0]hexane 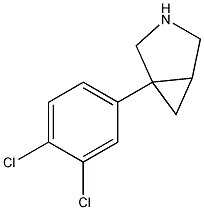
aka DOV 21,947. For some reason this bicyclic structures seems to crash the molecule viewer. Inhibits the reuptake of serotonin, norepinephrine, and dopamine (IC50 values of 12, 23, and 96 nM, respectively). DOV 21,947 reduces the duration of immobility in the forced swim test (using rats) with an oral minimum effective dose of 5 mg/kg. This antidepressant-like effect manifests in the absence of significant increases in motor activity at doses of up to 20 mg/kg. DOV 21,947 also produces a dose-dependent reduction in immobility in the tail suspension test, with a minimum effective oral dose of 5 mg/kg. Although this may not be a locomotor stimulant it looks like an interesting / promising antidepressant, also it has the similar structural features of this thread.
Antidepressant-like actions of DOV 21,947: a "triple" reuptake inhibitorPhil Skolnicka, Piotr Popikb, Aaron Janowskyc, Bernard Beera, Arnold S. Lippa,
European Journal of Pharmacology. 461, 99–104 (2003)DOI:
10.1016/S0014-2999(03)01310-4

______ _____ ____ ___ __ _
As a side note: there seem many stimulant molecules where the 4-halogen or the 3,4-halogen on the phenyl confers added potency. 4-fluoroamphetamine seems to be an interesting variation on amphetamine with slightly more sertonergic character. I wonder if 3,4-fluoroamphetamine would be even more serotonergically interesting. Which brings to mind (getting totally off track,) I think 3-methoxy-4-ethoxyamphetamine would be like MDA / MDMA. Look at pihkal 123 (MEPEA, 3-methoxy-4-ethoxyphenethylamine, lightly active at 300 mg "The effects were very quiet, very pleasant, and very light. There was nothing psychedelic here, but rather a gentle lifting of spirits") compared to pihkal 115 (MDPEA, 3,4-methylenedioxyphenethylamine, inactive at 300 mg). It stands to reason that the already-active MEPEA would become more potent and empathogenic effects accentuated as it followed the analogous path from MDPEA to MDA to MDMA. Also ME (metaescaline, 3,4-dimethoxy-5-ethoxyphenethylamine) seems to exhibit some "empathogenic" qualities, maybe the 3-ethoxy-4-methoxy-amphetamine would also. Additional interesting to look at would be MEA 3-methoxy-4-ethylamphetamine, similar to 3-methoxy-4-methylamphetamine MMA, and 3-methoxy-4-bromoamphetamine (with the last few, have to be careful of PMA trouble or PCA neurotoxicity of course).
______ _____ ____ ___ __ _
N-ethyl-3-phenylbicyclo[2.2.1]heptan-2-amine 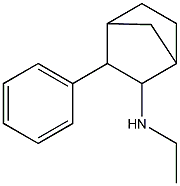
aka Fencamfamine. From the Hive "I have heard from a talented friend of mine who has exceptional taste that fencamfamine is his favorite stimulant of them all..." and another post mentioned "Fencamfamine (FCF) is a CNS stimulant that works by blockading dopamine reuptake and it has a potency similiar to amphetamine. Well that's what i was told by my coach who sold it to me, lol! I took a 0.5g IV dose for a research experiment once, that produced a very clean & subtle buzz which lasted for about 4-5 hours from memory. Nothing special, but I found sex was far more pleasurable using FCF as opposed to rooting on meth." Adding halogen 4 or 3,4 on the phenyl may further increase potency.
methyl 3-(4-bromophenyl)-8-methyl-8-aza-bicyclo[3.2.1]octane-2-carboxylate 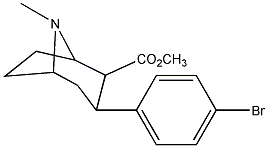
aka RTI-51 (para-iodo is RTI-55, equally potent). These are basically the most potent 3-phenyltropanes, with description of effects very similar to indatraline (very long lasting (16 h), very potent, reinforcing, locomotor stimulant, and substitute for cocaine). "During the first 2 h of behavioral observation that followed the intraperitoneal (IP) administration of the compound, each of the 3-phenyltropane analogs and cocaine increased horizontal activity). Cocaine, at 30.0 and 56.0 mg/kg, produced significantly greater activity than saline, with a peak effect at approximately 36 000 counts; RTI-51, at 0.49 mg/kg, significantly increased activity to 51 000 counts. RTI-51 (0.49 mg/kg) had an AUC significantly greater than that of 3.0 mg/kg cocaine. When the peak of the activity time course of the phenyltropane analogs were compared with that of cocaine, only 0.49 mg/kg RTI-51 produced a significantly greater peak effect than 3.0 mg/kg cocaine did. In rats, RTI-55 increased extracellular dopamine levels in the nucleus accumbens to about 350% of control values."
Molecule:
R-1-(Benzofuran-2-yl)-2-propylaminopentane ("CCC[C@@H](NCCC)CC2=CC1=CC=CC=C1O2")
aka (-)-BPAP. This compound is extremely interesting, has a number of unique activities and all of them positive. Also quite potent. Is not only catecholaminergic and serotonergic activity enhancer, but also a norepinephrine and dopamine uptake inhibitor and a weak serotonin uptake inhibitor, without adverse MAOI hypertensive crisis potential. [3H]dopamine, [3H]norepinephrine, and [3H]serotonin uptake IC50 were 42 nM, 52 nM, and 640 nM respectively; thus the effects of (-)-BPAP on dopamine and norepinephrine uptake were more potent than those of cocaine, while its potency at inhibiting serotonin uptake was weaker than that of cocaine. The uptake inhibitory effect of (-)-BPAP may be involved in motor stimulant effects in addition to its Catecholamine Enhancing and Serotonin Enhancing effect. In normal non-habituated rats, BPAP HCl increased motor activity during the entire 2-h observation period.
Post 211596 (missing)
(Rhodium: "BPAP, a catecholamine activity enhancer", General Discourse)
Molecule:
7-benzyl-1-ethyl, 4-oxo 1,8-naphthyridine-3-carboxylic acid ("O=C(C1=CN(CC)C2=C(C=CC(CC3=CC=CC=C3)=N2)C1=O)O")
aka amfonelic acid; cocaine-like stimulant with apparently very euphoric or addictive qualities. The rats go absolutely nuts for this one. Interesting that this stimulant looks unlike most other stimulant families, although still has phenalkylamine skeleton. In fact it is more closely related to antibacterial compounds. Should be pretty potent in humans, maybe 10 mg or less.
DA, NE, 5HT Uptake Inhibition: 7.2 nM, 41.1 nM, 879 nM1-(1-(benzothiophen-2-yl)cyclohexyl)piperidine 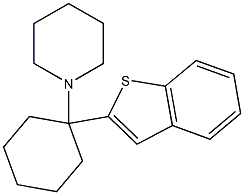
aka BTCP. This is a PCP analog but instead of being a dissociative it is a very specific cocaine-like stimulant compound, like amfonelic acid. The rats love this compound too. However, there are some health problems observed after repeated i.p. injections. In vivo Potency is comparable to cocaine; active metabolites may contribute to in vivo effect. "The reinforcing actions of BTCP were compared to those of cocaine at equimolar concentrations in drug-naive rats. Two doses (0.125 and 0.25 mg/infusion) that produced reliable self-administration in all the animals for cocaine and BTCP were then tested on a progressive-ratio schedule. At each dose, BTCP supported higher breaking points (BPs) than cocaine. The results demonstrate that rats readily acquire responding maintained by BTCP and suggest that BTCP may have greater reinforcing effects than cocaine at equimolar concentrations."
DA-uptake inhibition: IC50 11 nM vs. 296 nM for cocaineFurther Studies of the Structure-Activity Relationships of 1-[1-(2-Benzothienyl)cyclohexyl]piperidine; Synthesis and Evaluation of 1-(2-Benzothienyl)-N,N-dialkylcyclohexylamines at Dopamine Uptake and Phencyclidine Binding SitesXiao-shu He, Lionel P. Ramon, Mariena V. Mattson, Mohyee E. Eldefrawi and Brian R. de Costa,
J. Med. Chem. 36(25), 4075-4081 (1993)
Also
Post 99112 (missing)
(beagle boy: "Re: PCP Synthesis", Chemistry Discourse) beagle boy said: "The cpd was synthesized (not by me of course) and bioassayed by vaporizing and inhaling it in very small quantities (again, not by me). No effects were apparent, but in retrospect the amounts assayed were quite small, maybe on the order of 10-20 mgs or so."
Post 435648
(pHarmacist: "BTCP - a compound with cocaine-like properties", Novel Discourse) Two nice refs by pHarmacist
Post 507205
(7is: "More PCP related studies", Methods Discourse) Some more nice refs by 7is
1-(benzothiophen-2-yl)-N-propylcyclohexanamine 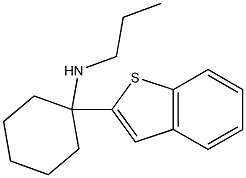
This BTCP analog might be about 2x more potent than BTCP based on binding and DAT inhibition.
DA-uptake inhibition: IC50 5 nMI have some more relevant references for these notes but I am too tired (read lazy) to retrieve them, check if they've been posted already, upload them, type up the reference in the proper format, and place them next to the appropriate location. This post is, even without them, a spacewasting bewhhymouth.
If references are desired, I'll do what I can, if I can, when I can, and where I can (probably a subsequent post). Which, by the way, I live in a can -- and can openers are the definite alphanumerical warnings. Which is to say, watch out for metallic indentations, the sky might crack open and blaze terrible light at any moment. What they didn't tell you about judgement day is that you get et.