Procedure for the Homogeneous Catalytic Hydrogenation of alpha,beta-Unsaturated Nitro Compounds using Triarylphosphine-Rhodium ComplexesRobert E. Harmon, Jack L. Parsons, and S. K. GuptaOrganic Preparations and Procedures 2(1), 25-27 (1970)In recent years the homogeneous hydrogenation of carbon-carbon double bond in the presence of other reducible functional groups such as carboxylic acids, esters, aldehydes, ketones, nitriles, etc. has received increased attention
1-5. The selective catalytic hydrogenation of an olefin in the presence of a nitro group is especially difficult. In this paper, we wish to describe a detailed procedure for the homogeneous hydrogenation of alpha,beta-unsaturated nitro compounds using tris(triphenylphosphine)chlororhodium(I) (
1)
1 and trichlorotris(4-biphenylyl-1-naphthylphenylphosphine)rhodium(III) (
2)
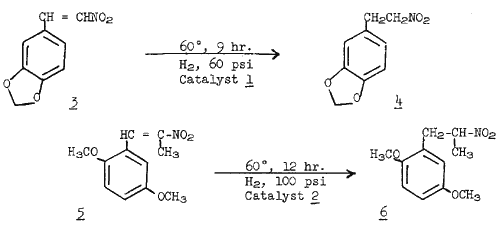
6. For instance, the hydrogenation of 3,4-methylenedioxy-ß-nitrostyrene (
3) using the catalyst
1, afforded a 84% yield of 2-(3,4-methylenedioxyphenyl)-nitroethane (
4). Similarly, the hydrogenation nitrostyrene (
5) gave a 90% yield of 2-(2,5-dimethoxyphenyl)-2-nitropropane (
6). Under identical reaction conditions (temperature 50°C, hydrogen gas pressure 80 psi, and reaction time 8 hr.), the catalyst
2 was found to be as effective as the well known Wilkinson's catalyst,
1.
 Experimental
Experimental3,4-Methylenedioxy-ß-nitrostyrene (
3) was prepared by the condensation of piperonal and nitromethane according to the procedure of Lange and Hambourger
7. 2,5-Dimethoxy-ß-methyl-ß-nitrostyrene (
5) is commercially available. Tris(triphenylphosphine)chlororhodium(I) was prepared by the reaction of an excess of triphenylphosphine with rhodium trichloride trihydrate according the procedure of Wilkinson and co-workers
3. Trichlorotris(4-biphenylyl-1-naphthylphenylphosphine)rhodium(III) was prepared by the reaction of 4-biphenylyl-1-naphthylphenylphosphine with rhodium trichloride trihydrate
2.
All solvents were deoxygenated by refluxing under a stream of argon for 3 hours, followed by storage under argon. Melting points were taken on a Thomas-Hoover melting point apparatus and are corrected. The hydrogenations were performed in a medium pressure hydrogenation apparatus.
The Hydrogenation of 3,4-methylenedioxy-ß-nitrostyrene (3) The compound 3 (2.5 g, 13.0 mmol) and tris(triphenylphosphine)chlororhodium(I) (
1) (0.40 g, 0.44 mmol) were dissolved in 300 ml of benzene under an argon atmosphere. The solution was transferred to the medium pressure reaction vessel and flushed with hydrogen gas 5 times. The reaction mixture was stirred for 9 hours at 60°C under a hydrogen gas pressure of 60 psi. After completion, the benzene was evaporated under reduced pressure. The oily residue was mixed with 50 ml of ethyl ether and filtered to remove the insoluble catalyst. The filtrate was evaporated to dryness. The residue was redissolved in ether, filtered, and the ether removed to give 2.1 g (84%) of 2-(3,4-methylenedioxyphenyl)-nitroethane (
4), mp 53-54°C.
The Hydrogenation of 2,5-Dimethoxy-ß-methyl-ß-nitrostyrene (5)The compound (3.0 g, 13.4 mmol) and trichlorotris (4-biphenylyl-1-naphthylphenylphosphine)rhodium (III) (0.40 g, 0.29 mmol) were dissolved in 300 ml of benzene-ethanol (1:1) under an argon atmosphere. The resulting clear red solution was transferred to the reaction vessel and after flushing 5 times with hydrogen gas, stirred for 12 hours at 60°C under a hydrogen gas pressure of 100 psi. Then the solvent was removed under reduced pressure and the remaining oil vacuum distilled to give 2.7 g (90%) of 2-(2,5-dimethoxyphenyl)-2-methyl-nitroethane, bp 135-140°C (0.1 mm).
References[1] J. A. Osborn, F. H. Jardine, J. F. Young, and G. Wilkinson,
J. Chem. Soc., A, 1711 (1966)[2] A. J. Birch and K. A. M. Walder,
J. Chem. Soc., C, 1894 (1966) [3] C. Djerassi, J. Gutzwiller,
J. Amer. Chem. Soc., 88, 4537 (1966)[4] F. H. Jardine, J. A Osborn, and G. Wilkinson,
J. Chem. Soc. A, 1574 (1967)[5] A. S. Hussey and Y. Takeuchi,
J. Amer. Chem. Soc., 21, 672 (1969)[6] R. E. Harmon, J. L. Parsons, and S. K. Gupta,
Organic Preparations and Procedures, this issue[7] N. Lange and N. Hambourger,
J. Amer. Chem. Soc., 53, 3865 (1931)