Novel Agonists of 5HT2C Receptors
Synthesis and Biological Evaluation of Substituted 2-(Indol-1-yl)-1-methylethylamines and 2-(Indeno[1,2-b]pyrrol-1-yl)-1-methylethylamines
Improved Therapeutics for Obsessive Compulsive DisorderMichael Bös, Francois Jenck, James R. Martin, Jean-Luc Moreau, Andrew J. Sleight, Jürgen Wichmann, and Ulrich WidmerJ. Med. Chem. 1997, 40, 2762-2769DOI:
10.1021/jm970030l

AbstractThe syntheses of a series of substituted 2-(indol-1-yl)-1-methylethylamines and 2-(indeno[1,2-b]pyrrol-1-yl)-1-methylethylamines are reported. The binding affinities of the compounds at 5HT
2C and 5HT
2A receptors (79% homology in the transmembrane domain) were determined. The ligands displayed selectivity for 5HT
2C receptors relative to 5HT
2A receptors. Compounds were functionally characterized both in vitro and in vivo as 5HT
2C receptor agonists.
5f,
5l,
5n,
5o,
5q,
14c,
14f,
14k, and
14m exhibited anticompulsive activity in an animal model of obsessive compulsive disorder.
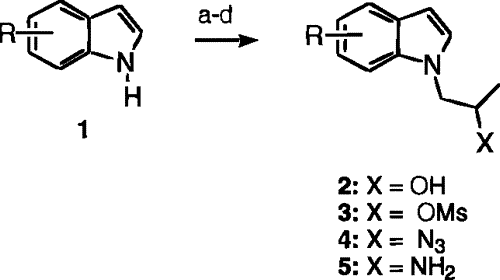 Scheme 1
Scheme 1 (a) Propylene oxide, NaH, THF; (b) MsCl, NEt
3, CH
2Cl
2; (c) NaN
3, DMF; (d) PtO
2, H
2, EtOH.
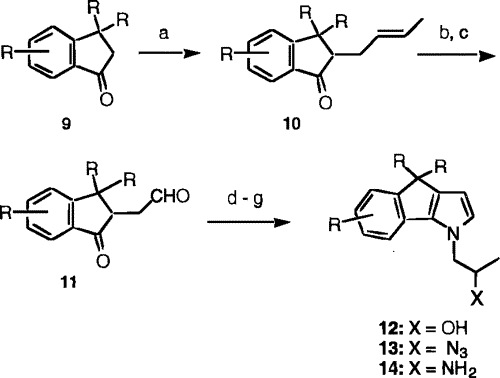 Scheme 3
Scheme 3 (a) 3-Buten-2-ol, p-TsA, 2,2-dimethoxypropane; (b) ozone, CH
2Cl
2-MeOH; (c) TFA, CH
2Cl
2; (d) 1-amino-2-propanol, p-TsA, toluene; (e) MsCl, NEt
3, CH
2Cl
2; (f) NaN
3, DMF; (g) PtO
2, H
2, EtOH.