101 things to do with used styrofoam cups: something to tickle your dendrites : )
Here's a little flowchart based on phenylpropanoids, starting from the addition products of styrene and acetic anhydride.
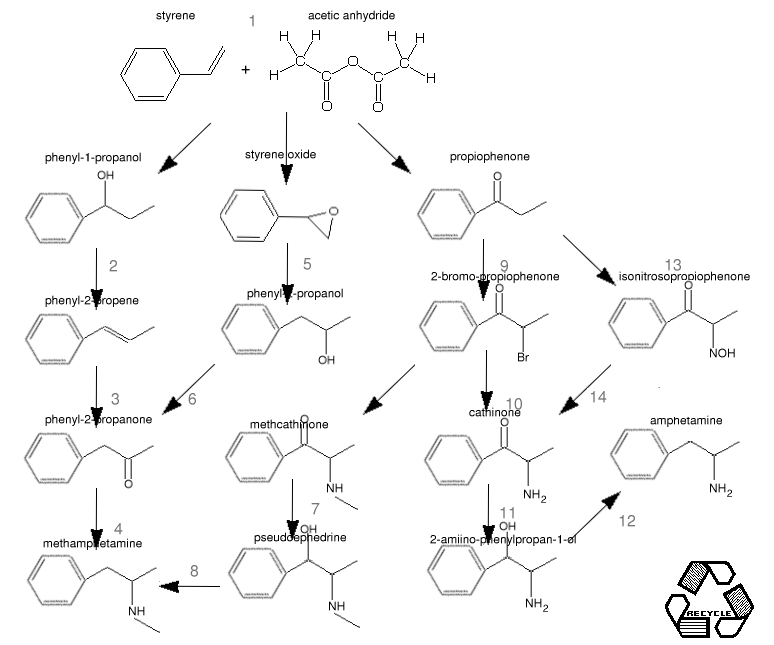
Some of the references for these are :
1 US PAT 3806548 1 phenylpropanol from styrene
2 J. Org. Chem 45(5) 917-919(1980)
None
(
https://www.thevespiary.org/rhodium/Rhodium/pdf/cuso4.dehydration.pdf)or None
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/propiopropen.html)
3
peracid oxidation
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/peracid.html)
4
reductive amination
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/alhg.osmium.html)of your choice.
5
Grignard.
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/p2pol.styrenepoxide.html)
6 Na2Cr2O7 oxidation; see
end of this document
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/p2p.oxymercuration.html).
7 [NaBH4 reduction, or reduction of your choice]
8 [NaBH4 reduction or reduction of your choice]
9 [
Post 423945
(Kinetic: "4-Methyl Methcathinone", Novel Discourse)Equimolar addition [/url]
10 Simple [
Post 423945
(Kinetic: "4-Methyl Methcathinone", Novel Discourse)amine substitution.[/url]
11
Reduction.
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/amph.urushibara.html)
12 Reduction of your choice, eg
CTH
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/cth.txt)
13 Org. Syn:. : Prep of isonitrosopropiophenone, or
Clayfen nitrosification with MW irradiation
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/mw.nitrostyrene.txt)
14 oxime reduction of your choice-
tthis one's nice
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/oxime2amine.zn-cu.html)because it worked where Hg/Al amalgum and Raney nickel didn't.
There are other ways to achieve the same result, I've just picked those which appeal to me.
Now what I'm really wanting to pick your brains about is this:
[1]Both techniques starting from propiophenone (9: Bromine addition, or 13: Clayfen nitritrosification/Org.Syn w. methyl nitrite) take advantage of the increased acidity of the carbon adjacent to the carbonyl group in propiophenone.Someone is interested (as I'm sure many would bee) in identifying a mechanism which goes directly from the phenone to the cathinone or methcathinone; for example, by catalysing substitution of a proton with the respective amine. Using basic conditions the acidic character of this carbon will be more pronounced. Instead of actually bonding a halide to the 2 carbon, only to remove it again, I wonder if the scant presence of a halide might be persuaded to nudge off a proton long enough for the amine to supply an electron: For example,
IF
a) the amine to be added is adsorbed onto anhydrous, ball-milled Lewis acid activated clay;
b) The reaction is done in acetone/hexane with AlCl
3 /CaO;
c) the reactants are MW irradiated.
d) a tiny amount of H
2O is present to provide that scant chloride presence (which will be controlled by CaO before doing any damage).
Do you have any comments on this?
Exactly how "acidic" is the 2 carbon in propiophenone? Maybe an amine is a bit ambitious a goal, perhaps an imime, but I am going to explore the activity of this site further. The Clayfen nitrosification reaction, under MW irradiation occurs in minutes- so there is definitely something to work with imho.
 [2]
[2]The yields from 1 are phenylpropanol:styrene epoxide:propiophenone 30%:28%:12.3% respectively.
Another route employing a peracid produces 48.7%:24.3%:27% respectively.
The second is more productive but slightly more complex.
Does anyone have any
bright ideas on how to favour the production of a particular product in this kind of reaction? Eg
a)by supplying an abundance of anhydride
b)the presence of peracid, peroxide or even MnO
2c)Presence of bases or other solvents.
Any clues on steering this type of reaction?
[3]Finally, would bisulfite adduct extraction work with the aqueous/aq. alcoholic mixture of the products of 1 for extraction of the ketone?
Hope you like the picture. It's part of a larger piece called
"The Nitrogen Cycle"
(http://elmhurst.edu/~chm/onlcourse/chm110/outlines/nitrogencycle.html). 