A versatile synthetic methodology for the synthesis of tryptopholsSimon J. Garden, , Rosângela B. da Silva and Angelo C. PintoTetrahedron, 58(42), 8399-8412 (2002)
(
https://www.thevespiary.org/rhodium/Rhodium/pdf/isatin2tryptophol.pdf)
DOI:
10.1016/S0040-4020(02)01048-7
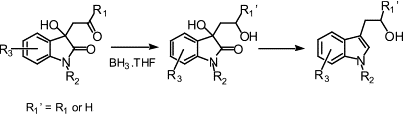
AbstractTryptophols have been obtained in high yields by the reduction of 3-substituted-dioxindoles (obtained by the aldol condensation reaction of ketones with isatins or by a modified Knovenagel malonate condensation) using a borane tetrahydrofuran complex. The reported methodology offers distinct advantages over existing methods for the synthesis of these compounds, including consistently greater yields, diastereoselective syntheses and the possibility for the synthesis of a wide range of structurally different tryptophols. The reduction reaction was found to proceed via an intermediate 1,3-diol-oxindole, which was obtained diastereoselectively and, which was subsequently reduced to the corresponding tryptophol.
Graphical AbstractA diverse range of tryptophols can be readily prepared by the BH
3·THF reduction of dioxindole derivatives. Intermediate diols are produced by diastereoselective reduction reaction.
Author Keywords: isatin; reduction; borane tetrahydrofuran complex; diastereoselective reduction; tryptophol