I recently found an article which might be of interest to fellow bees, because it might open a new route to 2-alkoxy-4,5-methylenedioxybenzaldehydes. MMDA-2 and its Tweetio analogue EMDA-2 (both described in
PiHKAL #133
(
http://www.erowid.org/library/books_online/pihkal/pihkal133.shtml)) are the end products I had in mind.
I'd be interested in pursuing its synthesis following the method(s) I have in mind. However, Fatum is not my partner in crime these days and a lack of time necessitates me to abandon or dramatically postpone the project. I decided to communicate my findings with the Fellow Bees, in the hope there would be others interested in the project. Maybe they can add their theoretical comments, or in the most ideal circumstances, try the synthesis in their own lab.
PART I. Synthesis of 2-methoxy-4,5-methylenedioxycinnamic acidReference: CK Sehgal, PL Kachroo, RL Sharma, SC Taneja, KL Dhar, CK Atal. Synthesis of
cis and
trans 2-methoxy-4,5-methylenedioxycinnamoylpiperidide and revised structure of a new alkaloid from
Piper peepuloides. Phytochemistry 18 (1979) 1865-1867. DOI:
10.1016/0031-9422(79)83072-1
Synthesis of 4,5-methylenedioxycoumarin (ayapin)Esculetin (1 g) was dissolved in 90 mL dry acetone and 10 mL DMF. To this solution were added 25 g potassium carbonate and 10 mL CH
2I
2. The mixture was refluxed on a water bath for 40 hours. The product crystallized from EtOAc-petroleum ether (500 mg), mp 230°C (lit mp 232°) [2]. It analyzed for C
10H
6O
4.
Synthesis of 2-methoxy-4,5-methylenedioxy-trans-cinnamic acidA solution of ayapin (300 mg) in aqueous NaOH (60%) was warmed on a water bath until the coumarin was completely dissolved. To this was added DMS (3 mL) slowly with continuous stirring. The mixture was warmed at 50°C and stirred for 6 hours. The reaction mixture was then cooled to 0°C and acidified with diluted HCl. The precipitate was collected, dried and crystallization from acetone gave the cinnamic acid (200 mg), mp 244°C. It analyzed for C
11H
10O
5Synthesis of 2-methoxy-4,5-methylenedioxy-cis-cinnamic acid6,7-Methylenedioxycoumarin (450 mg) was dissolved in 20 mL dry THF. 500 mg of NaH (60%) dispersed in oil were added slowly to the stirred solution, the temperature being maintained at 25°C. MeI (3 mL) was then added and stirring continued for a further 4 hours. The mixture was cooled and 40 mL 0.5 N HCl was added. It was then extracted with EtOAc and the organic layer was washed and dried. After removal of the solvent, 300 mg of a mixture of cis- and trans-(4:1) 2-methoxy-4,5-methylenedioxycinnamic acid was obtained.
Notes on Part I.1. The author methylated the phenyl bound hydroxyl group that came available after hydrolysis of the coumarin to its corresponding cinnamic acid. I suppose that DMS and MeI can be substituted for diethyl sulfate and EtI, respectively. This should result in 2-ethoxy-4,5-methylenedioxycinnamic acid.
2. Esculetin... Hey, thank you GC_MS! Let's grab the chem supplier's catalogue and... only $100 for a 5 g quantity? Sounds OK, no? I guess not. That is why I included a limited literature search on the synthesis of esculetin.
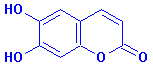
Esculetin, aesculetin, 6,7-dihydroxycoumarin: it's all the same for CAS 305-01-1, or:
Description of its synthesis using 1,2,4-triacetoxybenzene and hydrosuccinic acid:
- Amiard et al. Bull Soc Chim Fr (1947) 512
- Ishifuku et al. Yakugaku Zasshi 73 (1953) 332; CA.1954 2695
- HB Singh et al. Indian J Chem A 20(10) (1981) 1026
- YA Jackson. Heterocycles 41(9) (1995) 1979
Description of its synthesis using 3,4-dihydroxycinnamic acid (caffeic acid):
- F Borges et al. Helv Chim Acta 75(4) (1992) 1061 [45 days reaction time!]
- M Sato et al. Chem Pharm Bull 33(3) (1985) 1289
A final addition: 1,2,4-triacetoxybenzene is easily obtained via the Thiele-Winter acetoxylation of 1,4-benzoquinone (80%+ and even 95% with triflic acid catalyst). About the synthesis of 3,4-dihydroxycinnamic acid: I'd prepare it via the Perkin reaction of 3,4-dihydroxybenzaldehyde (demethylation of vanillin), or via a similar process (e.g. HOAc and NaBH
4 instead of AcO
2). I found one reference to an article describing the synthesis of caffeic acid, but I don't have access to it right now: J prakt Chem 145 (1936) 265.
PART II. Synthesis of 2-alkoxy-4,5-methylenedioxybenzaldehydeCinnamic acids can be oxidized to their corresponding benzaldehydes using KMnO
4/alumina. The procedure can be found on
Rh's site
(
https://www.thevespiary.org/rhodium/Rhodium/chemistry/benzaldehydes.kmno4.html), or you can take a look at the original article: S Lai et al. Synthesis (2001) 1644, DOI:
10.1055/s-2001-16760
.
Drawbacks: the article didn't test the cinnamic acids I discussed here, so there still is a chance that it won't work (for a reason I don't see). Also, the article works on a small scale. The article does work as described for some propenylbenzenes, but I only tested them on microscale as well.
Voila, that's it. Input - theoretical and practical - always welcome.