These are all snipets/articles from The Society for Amateur Scientists, which I take it, most Hive members are.
**************************************
Managing Mercury
Adapted from A Thousand and One Formulas by Sidney Gernsback
Mercury is difficult to manage, on account of its weight and the ease with which it picks up impurities. Stone-ware mortars are better for holding mercury than glass beakers, which easily crack when set down. all operations with mercury should be conducted over a tray or on a table with a groove around the edge.
Dust on the surface of Mercury may be removed by allowing the mercury to run through dry filter paper in which a few pin holes have been made. Or the mercury may be washed in a current of water, and then passed through a separating funnel. The little moisture on the surface may be easily removed with a blotting paper. Many metals easily dissolve in mercury; and a very little tin or zinc will spoil its fluidity. They are best removed by shaking the mercury in a bottle with a little nitric acid; the tin or zinc will then dissove, leaving the mercury pure.
**************************************
How to Clean Up Spilled Mercury
by Robert Dean
A lot of amateur scientists store and use mercury in their laboratories. If you're one of them, it's important that you understand the dangers associated with using mercury and that you are ready to clean up a spill should one happen.
Mercury, with its high surface tension and surprisingly high toxicity should not be used or stored where it might be mishandled. In a closed room with circulating air, spilled mercury can release enough vapor to injure anyone present. Mercury droplets cannot be swept up unless the brush is soaking wet. A film of water around a drop of mercury drags it to rest and prevents it from skating all over the floor. When the droplets can no longer skate they can be brushed together and disposed of with care. After disposal of the mercury, spread "flowers of sulfur" in all the cracks where mercury might be hiding. The finely divided elemental sulfur reacts with mercury vapor to form nonvolatile mercury sulfides. Do not let mercury get in touch with Zinc, Copper, Silver, or Gold because it can form amalgams with these and several other metals and remain on their surfaces. If that happens, this surface contamination will continue to release mercury vapor into the air. NEVER USE MERCURY OVER A CARPET! A spill there and the carpet must be pulled up and disposed of as contaminated waste.
[NOTE: Those of you who store mercury, please review how you're handling it. Just as every lab should have a first aid kit, any lab with stored liquid mercury inside should have a mercury clean up kit containing a plastic dust pan and brush, a mist bottle, and powered sulfur. You can use your SAS membership to purchase powdered sulfur at a discount just by letting Nancy know that you'd like some. SC.]
*************************************
Purifying and Working with Mercury
by John Strong
Excerpted from "Scientific American's The Amateur Scientist", first published in Procedures in Experimental Physics.
Although mercury approaches the noble metals in chemical inertness, it is easily contaminated, especially by other metals. This is because, as a liquid, it is a fairly good solvent. A simple test for the purity of a sample of mercury is to raise a clean glass rod slowly up through the metal surface. If the mercury is clean, the glass will come up without any adhering mercury droplets.
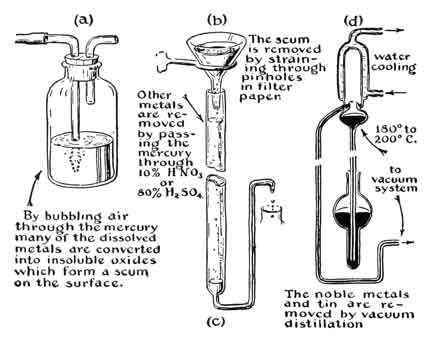
The contaminations commonly found in mercury may be classified according to the manner in which they can be easily removed. First come surface contaminations by materials which do not dissolve in the liquid metal and may, accordingly, be removed by filtering the metal through pinholes in filter paper or through a chamois skin. Second, there are the dissolved metals. Those which are oxidizable are first converted to insoluble oxides by the blowing of air through the mercury as shown in Fig. 9(a). The oxides form a scum on the mercury surface and may later be filtered off. Mercury is practically free of impurities of this type if, after air has been blown through the liquid metal for two hours, no scum has formed on the surface. The alkali metals fall into this class of impurities; here also belong zinc, with a high vapor pressure, and copper and lead, with low vapor pressures. These metals, which are more reactive than mercury, can also be removed by exposing the mercury to a solution of 10 per cent HNO3 or 80 per cent H2SO4. This is shown in Fig. 9(b). Thirdly, there are the dissolved metals, such as the noble metals and tin, which cannot be removed by oxidation or acid. Copper and lead may also be considered as belonging to this class of contaminations. These metals are removed by vacuum distillation of the mercury at a temperature of about 180° to 200°C. (at which temperature the mercury distills at the rate of approximately 1/2 g/cm2/sec.) as indicated by Fig. 9(c).
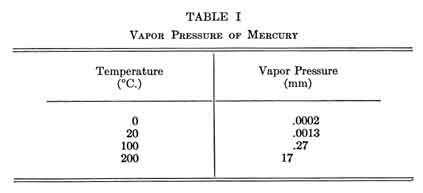
The vapor pressure of mercury is given in Table I. It is to be noted thoughtfully that at room temperature the vapor density of mercury is many times greater than the accepted nonpoisonous concentration limit, which is 1 milligram of mercury per cubic meter. According to Stock, continual breathing of air containing only 15 micrograms per cubic meter of mercury for a few weeks will make most persons ill. The vapor pressure of mercury is hazardously high in many laboratories. In a Berlin physical laboratory the typical concentration of mercury vapor in the air was found to be about 20 to 60 micrograms per cubic meter; in one room it was 500 micrograms per cubic meter. Heat produced by turning on mercury pumps doubled the concentration of mercury vapor in the air.
*************************************
LN2: Secrets of Zone Purification
by Shawn Carlson
One of the most useful ways to remove impurities from a liquid is to freeze it in a controlled way. Suppose, for instance, you want to purify some mercury you've got laying around. You could distill it, that is boil it and then condense the vapors. But since even a tiny concentration of mercury vapor is toxic, it's not a good thing to be boiling in your laboratory. But a good solution for the home scientist is to go to the other temperature at which mercury changes state, namely it's freezing point. As a general rule, a pure liquid freezes at a higher temperature than a contaminated one (that's why they sprinkle salt on roads in the winter time). So as heat is removed from a freezing liquid the pure molecules tend to bond together first, concentrating the impurities into the liquid that remains. So if you place a test tube of mercury into a bath of dry ice and alcohol, the mercury will freeze first around the inside of the tube, concentrating the impurities into the center. If you pour off the center fluid when about half of the volume is frozen, the mercury that remains will be substantially purer than what you started with.