Isocoumarins: Part IIIc Synthesis of Methyl- & Methylenedioxydihydroisocoumarins & a New Synthesis of (±)-5-MethylmelleinBhaskar H. Bhide & (Miss) Kashmira K. ShahIndian Journal of Chemistry, 19B, 9-12 (1980)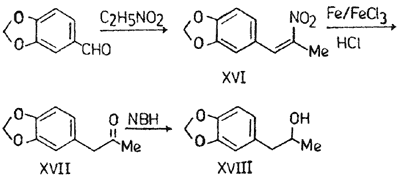 2-Nitro-1-(3,4-methylenedioxyphenyl)-1-propene (XVI)
2-Nitro-1-(3,4-methylenedioxyphenyl)-1-propene (XVI) Piperonal (35 g), nitroethane (50 ml), ammonium acetate (14 g) and glacial acetic acid (150 ml) were mixed and refluxed for 4 to 5 hr. The reaction mixture was then poured into ice-cold water. The fine golden yellow crystals of
XVI obtained were recrystallised from rectified spirit, mp 97-98°C; 37.2 g (77%).
1-(3',4'-Methylenedioxyphenyl)-2-propanone (XVII)*XVI (20 g) was dissolved in hot water (200 ml) and ethanol (200 ml). Iron powder (40 g), ferric chloride (1.6 g) and conc. HCl (10N, 24 ml) were added to it very slowly with constant heating and stirring. The mixture was refluxed with continuous stirring for 6 to 8 hr and concentrated to half the volume. The precipitated black iron oxide was filtered and washed with hot water and benzene. The combined washings were extracted with benzene and dried (Na
2SO
4).
XVII was obtained as brown oil after distilling benzene, and purified by distillation, bp 283-85°C; 10g (59%); oxime derivative, mp 86-87°C; DNP derivative, mp 138°C.
beta-Oxy-alpha-(3',4'-methylenedioxyphenyl)-propane (XVIII) NaBH
4 (5 g) was added slowly with stirring to a solution of
XVII (4.3 g) in methanol (50 ml). The mixture was stirred for 16 hr. Water was added and the solution extracted with CH
2Cl
2. After drying (Na
2SO
4) it was distilled to give
XVIII as dark brown oil which was purified by distillation under reduced pressure, bp 125°C/25mmHg, yellowish oil; 3.624 g (83.45%).
* J.M. Pepper, M. Saha, Can. J. Chem. 42, 13 (1964)