PCC Oxidation of Organoboranes Obtained Using AcetoxyborohydrideR.S. Dhillon, A.P. Kaur & G. KaurJ. Ind. Chem. Soc. 77, 453-454 (2000)
(
https://www.thevespiary.org/rhodium/Rhodium/pdf/safrole-mdp3p.pcc.borohydride.pdf)
SummaryAlkenes are hydroborated using sodium triacetoxyborohydride
1. The resulting organoborane is oxidized
2 with PCC
3, to form the corresponding aldehyde or ketone in 80% yield.
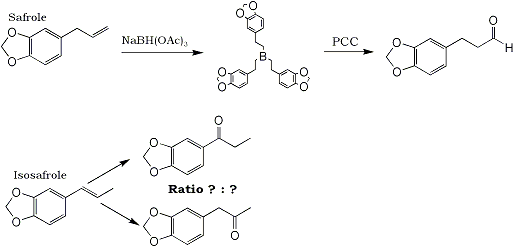
In Example #3, safrole subjected to the above one-pot reaction gives 3-(3,4-Methylenedioxyphenyl)-propionaldehyde ("MDP3P") in 81% yield.
My question is: What if isosafrole was used as the starting material instead of the safrole? In what proportions would the resulting ketones (MDP2P and MDP1P
4) form? Will one of them form in a significantly higher amount than the other? This procedure could very likely become an excellent method of preparation of one of those ketones. Someone will just have to test the reaction and report back their analysis of their reaction product(s).
 Notes:[1]
Notes:[1] In the paper it is prepared from NaBH
4/Hg(OAc)
2/THF, but I strongly reccommend this instead:
Post 439071
(Barium: "Prep of Sodium Triacetoxyborohydride", Novel Discourse)[2] PCC is not absolutely required for this oxidation, as I have seen CrO
3, as well as some peroxy-oxidant (H
2O
2 or Oxone) being used for the oxidation of organoboranes to ketones before. References upon request if you intend to try the reaction.
[3] Pyridine Chlorochromate
[4] MDP2P = 1-(3,4-Methylenedioxyphenyl)-2-propanone, MDMA precursor. MDP1P = 3,4-Methylenedioxypropiophenone, Methylone precursor.