Oxidative rearrangements of arylalkenes with [hydroxy(tosyloxy)iodo]benzene in 95% methanol: a general, regiospecific synthesis of alpha-aryl ketonesMichael W. Justik and Gerald F. KoserTet. Lett., 2004, 45(32), 6159-6163DOI:
10.1016/j.tetlet.2004.06.029
Fulltext:
Post 531247
(psychokitty: "New synthesis of phenylacetones", Novel Discourse)Abstract: The treatment of arylalkenes with [hydroxy(tosyloxy)iodo]benzene in 95% methanol affords the corresponding
alpha-aryl ketones. This oxidative rearrangement is general for acyclic and cyclic arylalkenes and permits regioselective syntheses of isomeric
alpha-phenyl ketone pairs.
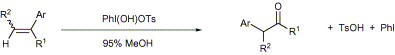
Synthesis of [hydroxy(tosyloxy)iodo]benzene:
Hypervalent Organoiodine. Reactions of Silver Arylsulfonates with Iodosobenzene DichlorideGerald F. Koser, Richard H. WettachJ. Org. Chem., 1977, 42, 1476-1478
Iodosobenzene diacetate (
1)
Reaction of
1 with p-Toluenesulfonic Acid in CH
3CN. p-Toluenesulfonic acid monohydrate (7.61 g, 40 mmol), dissolved in a minimum quantity of CH
3CN, was added to a solution/suspension of 1 (6.44 g, 20 mmol) in CH
3CN (45 mL) at room temperature. The reaction mixture immediately cleared and turned yellow and, upon standing at room temperature, gave phenylhydroxyltosyloxyiodine (
4a) as fine white needles with a yellowish hue. Two crops were isolated, yield 7.62 g (93%). The crude product was washed with acetone (removes p-toluenesulfonic acid) and ether (removes acetic acid) and dried: mp 135-138 °C
Synthesis of iodosobenzene diacetate
Easy Preparation of (Diacetoxyiodo)arenes from Iodoarenes with Sodium Percarbonate as the OxidantAgnieszka Zielinska and Lech SkulskiMolecules, 2002, 7(11), 806-809.pdf
(
http://www.mdpi.org/molecules/papers/71100806.pdf)
Sodium percarbonate (18.4 mmol, 330% excess) was slowly added portionwise to a stirred mixture of Ac
2O (7.0 mL), AcOH (5.8 mL), and CH
2Cl
2 (40 mL). The stirring was continued for 1.5 h at <= 30°C. An appropriate iodoarene was then added (6.4 mmol), and the reaction mixture was then stirred at 40°C for 5 h. After cooling, the precipitated CH
3COONa was collected by filtration under reduced pressure, washed with CH
2Cl
2 (2 x 15 mL) and discarded. The filtrates were evaporated under vacuum, and cold (0-5 °C) 10% aq. CH
3COOH (15 mL) was rapidly added. The flask was left in a cooler for a few hours. The colorless crystals formed were collected by filtration, washed with hexane and air-dried in the dark. Most of those crude products thus obtained need not be further purified, as they were 96-99% pure, according to iodometric analysis [6]. When necessary, the crude products were recrystallized from AcOEt/Ac
2O (9:1, v/v) [2]. They should be stored in the dark, preferably in a cooler. See Table 1 for more details. Under these conditions 4-iodotoluene and 4-chloroiodobenzene were unexpectedly oxidized to the corresponding iodylarenes (recrystallized from boiling water).
[2] Kazmierczak, P.; Skulski, L. A Simple, Two-Step Conversion of Various Iodoarenes to (Diacetoxyiodo)arenes with Chromium(VI) Oxide as the Oxidant. Synthesis 1998, 1721-1723, and references therein.
[6] Lucas, H. J.; Kennedy, E. R.; Formo, M. W. Org. Synth. 1942, 22, 70.
For a synthesis without acetic anhydride but with peracetic acid/acetic acid, see:
Organic Syntheses, CV 5, 660
(
http://www.orgsyn.org/orgsyn/prep.asp?prep=cv5p0660)