Rhodium-catalysed Isomerisation of Some Unsaturated Organic SubstratesJean Andrieux. Derek H. R. Barton. and Henri PatinJ. Chem. Soc. Perkin Trans. 1, 359-363 (1977)The versatile rhodium trichloride trihydrate has been employed in catalytic amounts to promote double-bond migration. This method allows good yields in otherwise difficult, or impossible, exocyclic-endocyclic isomerisations.
Migration of a double bond may be difficult to bring about when the molecule concerned is fragile. The classical methods usually require drastic conditions, i. e. strong bases or acids or high temperature heterogeneous catalysis
1. The recent discovery that several transition metal complexes, used as homogeneous catalysts, can isomerise double bonds in simple olefinic systems
2,3 has been applied to a few more sophisticated molecules
4,5,6. The potential interest of these reagents in organic chemistry lies in their selectivity and ability to bring about reaction under mild conditions.
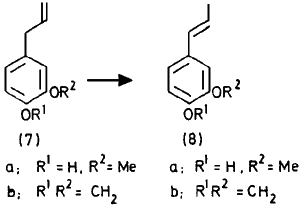
Isomerisation occurs readily when the migrating double bond moves into conjugation with an aromatic nucleus. In contrast to the fact that allyl groupings are isomerised by strong bases with formation of significant amounts of
cis-isomer
1, eugenol (
7a) and safrole (
7b) gave predominantly the
trans-isomers (
8a) and (
8b). The 1:9
cis-trans ratio in isoeugenol was altered neither by the amount of catalyst nor by the reaction time, but was slightly sensitive to an increase of the reaction temperature, as recently noted
18.
The commercially available isoeugenol contains approximately 40% of
cis-isomer. The
cis-trans ratio remained unchanged even after a long period of contact with the rhodium catalyst. It has been reported that the ratio of
cis- to
trans-isosafrole was 4:6 when
tris(triphenylphosphine)rhodium chloride was used as catalyst
5.
 Isomerisation of Eugenol
Isomerisation of Eugenol
To eugenol (20 g) in ethanol (5 ml) was added RhCl3·3 H20 (0.1 g). The temperature was maintained at 20°C by cooling and after 2 h the mixture was poured into water and extracted with ether. The usual work-up was followed by distillation; the yield of the fraction bp 140-142°C at 15 mmHg was 92%. The minimum amount of catalyst for 20 g of eugenol is 5 mg; complete transformation requires 24 h in this case. The amount of the trans-isomer of eugenol was always at least 90%.
Isomerisation of Safrole
Under the same conditions safrole (20 g) was converted into isosafrole, bp 130-132°C at 15 mmHg (93%). The amount of trans-isomer was about 80%.References[1] A. J. Hubert and H. Reimlinger,
"The Isomerization of Olefins Part I. Base-Catalysed Isomerization of Olefins",
Synthesis 97 (1969);
"The Isomerization of Olefins Part II. Thermal and Catalytic Isomerization of Olefins using Acids, Metals, Metal Complexes, or Boron Compounds as Catalysts",
Synthesis 405 (1970) Post 473104
(Rhodium: "Review: The Isomerization of Olefins, Part I & II", Novel Discourse)[2] G. C. Bond,
Ann. Reports, 63, 27 (1966)[3] R. Cramer,
"Transition metal catalysis exemplified by some rhodium-promoted reactions of olefins",
Acc. Chem. Res., 1, 186 (1968)[4] J. F. Biellmann and M. J. Jung,
"The mechanism of isomerization of an olefin and its possible relation to the mechanism of the catalytic hydrogenation with tris
(triphenylphosphine)rhodium chloride",
J. Amer. Chem. Soc., 90, 1673-74 (1958)[5] A. J. Birch and G. S. R. Subba Rao,
"Olefin isomerisations using tristriphenylphosphinerhodium chloride",
Tetrahedron Letters 3797 (1968) [6] E. J. Corey and J. W. Suggs,
"Selective cleavage of allyl ethers under mild conditions by transition metal reagents",
J. Org. Chem. 38, 3224 (1973);
"A new method for protection of carbonyl compounds as 5-methylene-1,3-dioxanes",
Tetrahedron Letters 3775 (1975) [18] Patent DE2508347