Benzene Hydrocarbons with a Pseudo Allyl Side Chain: Methovinylbenzene and its Homologues. Study of Certain Molecular Migrations. Second Part. Study of Molecular Transpositions which Accompany the Transformation of  -Glycols and their Derivatives into Aldehydes and Ketones.Tiffeneau, M.
-Glycols and their Derivatives into Aldehydes and Ketones.Tiffeneau, M.Ann. chim. phys., [8], Vol 10, pp 322-78 (1908)
(
https://www.thevespiary.org/rhodium/Rhodium/djvu/tiffeneau.carbures-benzenique.djvu)
AbstractThis paper consists of a continuation of the experimental study and theoretical consideration of certain migrations which take place in derivatives of glycols under the influence of various reagents. It was previously observed (Ann. chim. phys. [8], 10, 145-98) that halohydrins of  -glycols, when treated with alkalies and then distilled, gave first an oxide and then an aldehyde, thus but with AgNO3, or HgO a more deep-seated transformation took place, resulting in the migration of a hydrocarbon radicle. Thus, from the iodohydrin of methylphenylglycol, phenylacetone was obtained according to PhCOH(Me)CH2I
-glycols, when treated with alkalies and then distilled, gave first an oxide and then an aldehyde, thus but with AgNO3, or HgO a more deep-seated transformation took place, resulting in the migration of a hydrocarbon radicle. Thus, from the iodohydrin of methylphenylglycol, phenylacetone was obtained according to PhCOH(Me)CH2I  PhCH2COMe. The author has studied successively the glycols, the oxides of ethylene and the halohydrins.
PhCH2COMe. The author has studied successively the glycols, the oxides of ethylene and the halohydrins.Among the

-glycols he finds that the primary-secondary and primary-tertiary, after dehydration, rearrange themselves into aldehydes with only a shifting of the position of the hydrogen atom; for example, RCH(OH)CH
2OH

RCH=CHOH RCH
2COH; bi-secondary into ketones with only the migration of the hydrogen atom, e.g., MeCH(OH)CH(OH)Me

MeCH
2COMe, or into aldehyde, with wandering of the phenyl group, provided this group is attached to the carbon atom carrying hydroxyl, which does not enter into the dehydration, thus, MeCH=C(OH)Me and PhCH=C(OH)Me, isomerize without migration of hydrocarbon group. PhCH=C(OH)Ph isomerizes with migration of a hydrocarbon group. In the case of the bi-tertiary the rearrangement cannot take place without a wandering of a hydrocarbon radicle. All the oxides of ethylene, except the tetra-substituted derivatives, isomerize with the simple migration of the hydrogen atom.
In the case of the isomerization of iodohydrins of aromatic nature into aldehydes and ketones, the migration of the phenyl group takes place when it is attached in the hypothetical intermediate substance to the carbon carrying the hydroxyl group; the same is true of the magnesium derivatives of the halohydrins.
ExperimentalUnsymmetrical methylphenylethyleneglycol, PhMeC(OH)CH
2OH, can be made by the action of barium hydrate or carbonate in the presence of water on the dibromide of methovinylbenzene, or from methylmagnesium iodide and benzyl carbinol, m. 42-43°C, b
26 160-162°C. Heated with dilute H
2SO
4 it gives hydratropic aldehyde. Unsym. methyl-p-tolylethyleneglycol, MeC
6H
4COHMeCH
2OH, m. 36°C, b
15 175-80°C, obtained in a similar manner to the preceding compound, by boiling with dil. H
2SO
4 gives methylhydratropic aldehyde, b
730 219-221°C. Unsym. diphenylethyleneglycol, Ph
2C(OH)CH
2OH, from phenyl magnesium bromide and ethyl glycollate, m. 122°C. Boiled with dil. H
2SO
4, it gives diphenylacetaldehyde, b
15 170-175°C.
Symmetrical methylphenylglycol (See Fincke, Ber., 17, 710) gives phenylacetone.The oxide of styrene shows a remarkable stability in the presence of hydrolytic agents, since it does not give phenylacetaldehyde with H
2SO
4 and HNO
3 or AgNO
3; the oxide of methylvinylbenzene, on the other hand, isomerizes very rapidly by heating with dil. H
2SO
4.
p-Methoxystyrolene, obtained by the method of Klages (
Ber., 36, 3592) gives with mercuric oxide and iodine, methylphenylacetaldehyde, b. 255-256°C, d
0 = 1.140, oxime, m. 121°C (
Bouveault, Compt. rend., 135, 41), semicarbazone, m. 181-182°C. On reduction with zinc and acetic acid, the aldehyde gives the acetate of p-methoxyphenylethanol, b
11, 156-7°C, d
0= 1.101.
Phenylpropylene, by passing through the intermediate iodohydrin, gives hydratropic aldehyde, whose oxime is a liquid, b7 124°C. Reduced with zinc and acetic acid, the aldehyde gives the acetate of hydratropic alcohol, from which, by saponification, the alcohol may be obtained, b
14 113-4°C. Phenylmagnesium bromide, with the aldehyde, gives the alcohol PhCHMeCHOHPh. From the iodohydrin of phenylpropylene one obtains phenyl-1-dimethylamino-2-propanol-1 (
Fourneau, J. pharm. chim. [6], 20, 488), indicating the constitution of the iodohydrin. Methyl-3-phenylbutylene, obtained by dehydration of the alcohol, PhCHOHC
2CHMe
2, gives isopropylphenylacetaldehyde, b. 215-220°C, semicarbazone, m. 140°C.
The iodohydrin of methylphenylglycol, PhMeCOHCH2I, gives phenylacetone. This ketone with phenylmagnesium bromide gives

,
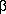
-diphenylpropylene (
Klages, Ber., 35, 2648).
n-Propylstyrolene, obtained from
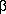
-propylcinnamic acid, which in turn was made from the ethyl ester, formed by the action of phenylbutanone in the presence of magnesium or ethyliodacetate, gives, through its iodohydrin, benzylpropylketone. The n-propylstyrolene gives a liquid dibromide.
Diphenylmethylcarbinol, Ph
2COHMe, m. 80-81°C (
Tiffeneau, Bull. soc. chim. [3], 27, 292; Klages, Ber., 25, 2646; Masson, Compt. rend., 135, 533) by distillation at the ordinary temperature, gives the unsaturated hydrocarbon, Ph
2CH=CH
2, which, through its iodohydrin, yield desoxybenzoin.
p-Tolylphenylmethyl carbinol, MeC
6H
4(Ph)C(OH)Me, is obtained as liquid from p-methylacetophenone. By repeated distillation at ordinary temperatures, it gives p-tolylphenylethylene, b. 285-286°C, b
6 145-146°C The latter yields p-tolylacetophenone. Phenyl-2-butylene, through its iodohydrin, gives methylbenzylmethyl ketone, PhCHMeCOMe, b. 210-212°C, b
23, 106-107°C.
Phenyl-2-n-amylene, PhMeC=CHCH
2Me, gives a ketone which does not combine with sodium sulphite, b. 225-227°C. Semicarbazone, m. 188°C. Phenyl-3-n-amylene, PhC(Et)=CHMe, was made by dehydration of the diethylphenylcarbinol made by action of ethyl magnesium bromide on ethylbenzoate, b. 197-9°C, d
0=0.9321. Through the iodohydrin it gives methylbenzylethyl ketone, MePhCHCOEt, b. 222-225°C, d
0=0.982. Semicarbazone, m. 172°C. By the action of ethyl magnesium bromide on chloracetophenone, benzylethylketone is formed, semicarbazone, m. 146°C.