J.Org.Chem., 63,9.1998;2858-2866
full article

very raw OCR'ed w/o pictures
 summary
summaryAlkylation of the aromatic nucleus, an important reaction in industry and synthetic organic chemistry, has traditionally been carried out by the well-known Friedel -Crafts reaction employing Lewis acid catalysts such as AlCl
3 and BF
, or by using highly reactive organometallic reagents. Although protic acids such as anhydrous HF and concentrated H
2S0
4 have also been used in the alkylation of the aromatic nucleus, the notoriously corrosive, highly toxic, and hazardous nature of these agents has precluded their common use under ordinary laboratory conditions. Various organic sulfonic acids have, on occasion, been used as catalysts in Friedel-Crafts alkylations, but to our knowledge the chemistry and the scope of these reactions for common laboratory use have never been exploited in detail. In the present study we have characterized commercially available p-toluenesulfonic acid monohydrate (TsOH) as an efficient catalyst for the intermolecular coupling of the aromatic nucleus with activated alkyl halides, alkenes, or tosylates under mild conditions in an open atmosphere. In comparison to conventional Friedel-Crafts catalysts such as AlCl
3, BF
3, HF, and concentrated H
2SO
4, the extent of the formation of undesired products from side reactions such as transalkylation, polymerization, etc. was minimal with the TsOH-catalyzed reaction. The ability to recover and reuse the catalyst from the reaction mixtures, minimal generation of environmentally unfriendly waste, high specificity of the reaction, and the low cost of the catalyst are important advantages of the TsOH catalyst over the other conventional Friedel-Crafts catalysts.
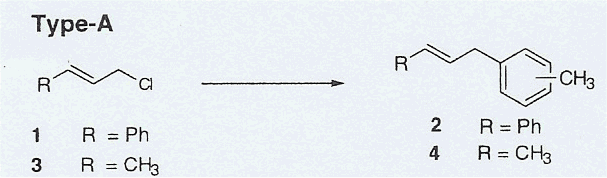
Alkylation of Toluene. A mixture of the desired alkyl substrate (0.05 mol) and p-toluenesulfonic acid monohydrate (11.4 g, 0.06 mol) in 53 mL of toluene (0.5 mol) was refluxed for the appropriate times indicated in Table 1, with magnetic stirring. The reaction mixture was cooled to room temperature, allowing the crystalline catalyst to separate. The solid catalyst was then filtered and washed with hexane (2 x 25 mL). The resultant organic filtrate was washed first with 0.1 M NaHCO
3 solution and then with distilled water and dried over anhydrous Na
2SO
4. The evaporation of the organic solvent in vacuo yielded crude product mixtures which were, subjected to further purification whenever it was appropriate.
--
psyloxy--